Chapter 10 States of Matter
750 likes | 1.6k Vues
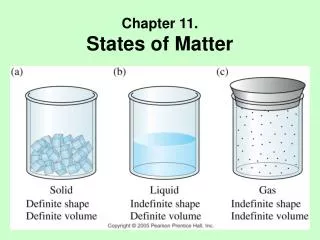
Chapter 10 States of Matter. 10.1 The Kinetic-Molecular Theory of Matter. Kinetic-Molecular Theory of Gases. Particles of matter are ALWAYS in motion; constant, rapid motion. (kinetic energy!) Particles are very small & relatively far apart.

Chapter 10 States of Matter
E N D
Presentation Transcript
Chapter 10States of Matter 10.1 The Kinetic-Molecular Theory of Matter
Kinetic-Molecular Theory of Gases • Particles of matter are ALWAYS in motion; constant, rapid motion. (kinetic energy!) • Particles are very small & relatively far apart. • Collisions of particles with container walls cause pressure exerted by gas. • Volume of individual particles is zero. • Particles exert no attractive or repulsive forces on each other.
Kinetic-Molecular Theory of Gases • Gas particles undergo elastic collisions: Collisions in which no energy is lost • Average kinetic energy is directly proportional to Kelvin temperature of a gas. Air Hockey Table
Ideal Gas • An imaginary gas that perfectly fits all the assumptions of the kinetic-molecular theory • A gas with its particles in constant random motion without attraction for each other is called an Ideal Gas. These particles undergo elastic collisions. • Nearly all real gases behave as ideal gases EXCEPT at very low temperatures or high pressures.
Real Gases • A gas that does not behave completely according to the assumptions of the kinetic-molecular theory. • Real gases occupy space and exert attractive forces on one another
Kinetic-Molecular Theory of the Nature of Gases • Expansion Gases do not have a definite shape or volume Gases take the shape of their containers Gases evenly distribute themselves within a container • Fluidity Gas particles easily flow past one another • Low Density • A substance in the gaseous state has 1/1000 the density of the same substance in the liquid or solid state • Compressibility • Gases can be compressed, decreasing the distance between particles, and decreasing the volume occupied by the gas
Kinetic-Molecular Theory of the Nature of Gases • Diffusion Spontaneous mixing of particles of two substances caused by their random motion • Rate of diffusion is dependent upon: • speed of particles • diameter of particles • attractive forces between the particles
Kinetic-Molecular Theory of the Nature of Gases • Effusion Process by which particles under pressure pass through a tiny opening • Rate of effusion is dependent upon: • speed of particles (small molecules have greater speed than large molecules at the same temperature, so the effuse more rapidly)
Chapter 10States of Matter 10.2 Liquids
Some Properties of a Liquid • Surface Tension: The resistance to an increase in its surface area (polar molecules, liquid metals). A force that tends to pull adjacent parts of a liquid's surface together, thereby decreasing surface area to the smallest possible size.
Some Properties of a Liquid • Capillary Action:Spontaneous rising of a liquid in a narrow tube.
Some Properties of a Liquid • Viscosity: Resistance to flow (molecules with large intermolecular forces).
Some Properties of Liquids Volatility • Liquids that have weak forces of attraction and evaporate easily Nonvolatile Liquids • Liquids that have strong forces of attraction and do not evaporate easily
Properties of Fluids Relative High Density • 10% less dense than solids (average) • Water is an exception • 1000x more dense than gases Relative Incompressibility • The volume of liquids doesn't change appreciably when pressure is applied Ability to Diffuse • Liquids diffuse and mix with other liquids • Rate of diffusion increases with temperature
Chapter 10States of Matter 10.3 Solids
Types of Solids • Crystalline Solids: highly regular arrangement of their components • [table salt (NaCl), pyrite (FeS2)].
Types of Solids • Amorphous solids aka supercooled liquids: considerable disorder in their structures (glass). • Greek for "without shape" • Formation of amorphous solids: • Rapid cooling of molten materials can prevent the formation of crystals * They do not have definite melting points
Representation of Components in a Crystalline Solid Lattice: A 3-dimensional system of points designating the centers of components (atoms, ions, or molecules) that make up the substance.
Types of Crystalline Solids Ionic Solid: contains ions at the points of the lattice that describe the structure of the solid (NaCl).
Unit Cell • The smallest portion of a crystal lattice that shows the three-dimensional pattern of the entire lattice
Types of Crystalline Solids Molecular Solid: discrete covalently bonded molecules at each of its lattice points (sucrose, ice).
Packing in Metals Model: Packing uniform, hard spheres to best use available space. This is called closest packing. Each atom has 12 nearest neighbors.
Metal Alloys • Substitutional Alloy: some metal atoms replaced by others of similar size. • brass = Cu/Zn
Metal Alloys(continued) • Interstitial Alloy:Interstices (holes) in closest packed metal structure are occupied by small atoms. steel = iron + carbon
Network Solids • Composed of strong directional covalent bondsthat are best viewed as a “giant molecule”. • brittle (non-flexible) • do not conduct heat or electricity • carbon, silicon-based • graphite, diamond, ceramics, glass
Chapter 10States of Matter 10.4 Changes of State
Equilibrium • Dynamic condition in which two opposing changes occur at equal rates in a closed system • A closed system at constant temperature will reach an equilibrium position at which the rates of evaporation and condensation will be the same
Equilibrium Vapor Pressure • The pressure of the vapor present at equilibrium. • Determined principally by the size of the intermolecular forces in the liquid. • Increases significantly with temperature. • Volatile liquidshave high vapor pressures. Increasing the temperature will move more particles into the vapor phase to compensate for the new energy
The conversion of a liquid to a vapor within the liquid as well as at its surface. It occurs when the equilibrium vapor pressure of the liquid equals the atmospheric pressure Boiling Boiling Point • The temperature at which the equilibrium vapor pressure of the liquid equals the atmospheric pressure Water boils at 100 °C at 1 atm pressure Water boils above 100 °C at higher pressures Water boils below 100 °C at lower pressures
LeChatelier’s Principle When a system at equilibrium is placed under stress, the system will undergo a change in such a way as to relieve that stress.
Translation: When you take something away from a system at equilibrium, the system shifts in such a way as to replace what you’ve taken away. When you add something to a system at equilibrium, the system shifts in such a way as to use up what you’ve added.
LeChatelier’s Example #1 A closed container of ice and water at equilibrium. The temperature is raised. Ice + Energy Water The equilibrium of the system shifts to the _______ to use up the added energy. right
LeChatelier’s Example #2 A closed container of N2O4 and NO2 at equilibrium. NO2 is added to the container. N2O4 + Energy 2 NO2 The equilibrium of the system shifts to the _______ to use up the added NO2. left
LeChatelier’s Example #3 A closed container of water and its vapor at equilibrium. Vapor is removed from the system. water + Energy vapor The equilibrium of the system shifts to the _______ to produce more vapor. right
constant Temperature remains __________ during a phase change. Water phase changes
Phase Diagram • Represents phases as a function of temperature and pressure. • Critical temperature: temperature above which the vapor can not be liquefied. • Critical pressure: pressure required to liquefy AT the critical temperature. • Critical point: critical temperature and pressure (for water, Tc = 374°C and 218 atm).
Water Water
Carbon dioxide Carbon dioxide
Carbon Carbon
Chapter 10States of Matter 10.5 Water
Sea Ice • Ice forms on top of the ocean in a thin layer & acts to insulate the warmer waters below from the colder air temperatures. • This occurs in the polar regions, the Artic & Antarctic • Since ice is less dense than liquid water, it will float on top, instead of sinking which would kill all life below the surface. • Sea ice is not the same as an iceberg. Icebergs are pieces of glaciers which are formed by snowfall on land. • Sea ice is not salty, as the hydrogen bonds that hold ice together will not form properly if salt remains in the structure.