Heat Transfer Modeling and Applications
1.02k likes | 1.4k Vues
Heat Transfer Modeling and Applications. A Short Course Reza Toossi, Ph.D, P.E. California State University, Long Beach. Outline. Scope and Types Energy Equation Formulation Conversion Mechanisms Dimensionless Parameters in Heat Transfer Modes of Heat Transfer

Heat Transfer Modeling and Applications
E N D
Presentation Transcript
Heat Transfer Modeling and Applications A Short Course Reza Toossi, Ph.D, P.E. California State University, Long Beach
Outline • Scope and Types • Energy Equation • Formulation • Conversion Mechanisms • Dimensionless Parameters in Heat Transfer • Modes of Heat Transfer • Conduction, Convection, and Radiation • Correlations • Combined Modes • Heat Transfer in Multiple Phases • Conjugate Heat Transfer • Composites • Phase Change
Scales • T 0 K – 1.4x1032 K • L 1.6x10-35 m – 1.6x1026 m (15 bly) • t 5.4x10-44 s – 2.7x1017 s • m 10-69 kg - 1054 kg
Mechanical Systems • Mechanically determined problems • Can be solved only by Newton’s Law of Motion and Conservation of Mass Examples: • Free fall of a body; F = ma • Dynamics of rigid bodies in absence of friction • Flow of ideal fluids between two parallel plates • Mechanically undetermined problems • Additional laws are needed Examples: • Dynamics of rigid bodies with friction • Dynamics of deformable body
Thermal Systems • Thermodynamically determined problems • Can be solved by the general laws of mechanics and the first and second laws of thermodynamics. Example: • Flow of steady 1-D isentropic and subsonic fluid through a nozzle. mass d(rAV) = 0 Momentum dp+ rAdV = 0 Energy du +pd(1/r) = 0 • Thermodynamically undetermined problems • Heat Transfer (modes of heat transfer) • Gas Dynamics (equation of state)
Knudsen Number • Continuum regime (Kn < 0.01) • Slip flow regime (0.01 < Kn < 0.1) • Transition regime (0.1 < Kn < 3) • Free molecular flow regime (Kn > 3)
Formulations • Differential • Integral • Integral-Differential • Thermal Nodes
Differential-Volume Fire Walking Infinite flat plate Solution: Interface Temperature (no contact resistance)
Integral Volume (lumped) • Heat losses under the condition of hypothermia Take cvV = 5x105 J/K, Sv = 400 W, and QA= Qr = 800 W; T = 10oC Get t =3.47 hr
Surface Coating • Droplet Impingement on a Hot Surface • Temperature is uniform within the droplet (particle) • Particle temperature varies during its flight • Sensible heating until particle reaches its melting temperature. Melting occurs at T = Tsl • Time to reach melting temperature
Thermal Nodes - Slab - Cylinder - Sphere
Heat Source • Phase Change • Chemical Reaction • Nuclear Fission and Fusion • Surface Friction Heating • Viscous Heating • Ultrasound Heating • Microwave Heating • Joule Heating • Thermolectric Heating (and Cooling)
Chemical Reaction ar is pre-exponential factor = func (f)
Surface Friction Heating • Interfacial Energy Conversion of Mechanical Energy μF is the friction coefficient pc is the contact or joint pressure Δui is the interface relative velocity
Viscous Heating • Volumetric Conversion of Mechanical Energy Due to Fluid Viscosity Example: Viscous heating in a ball bearing. The bearing is 0.2 mm in diameter, and ui = 1 m/s. Engine oil at STP has a viscosity of mf = 0.366 Pa/s
Ultrasound Heating • Volumetric Conversion of Longitudinal Acoustic Waves to Thermal Energy sac acoustic absorption coefficient = 1.4 for blood = 14 for muscle = 31 for skin = 161 for bone as speed of sound = 1,519 m/s in tissues = 3,445 m/s in bones f frequency μ dynamic viscosity γ specific heat ratio g = cp/cv Pr Prandtl number
Microwave (Dielectric) Heating • Volumetric Conversion of Electromagnetic to Molecular Vibration (Heat) ee (V/m) electric field intensity eec dielectric loss factor (relative permittivity) e0 permittivity of free space f (Hz) oscillation frequency
Joule heating • Conversion of Electrical Energy to Heat
Thermoelectric Power Generation Unit • Direct Electrical Power Generation by • Heat Absorption at a Hot Junction • Rejecting the Peltier Heat at the Cold Junction • Bismuth-telluride cold p-n junction • S,p=230x10-6 V/k, S,n= -210x10-6 V/k, Je=10 A • The Peltier heat absorbed at the cold junction • Tc = 120oC Q = -1.73 W • Tc = 20oC Q = -1.29 W • Tc = - 80oC Q = -0.85 W
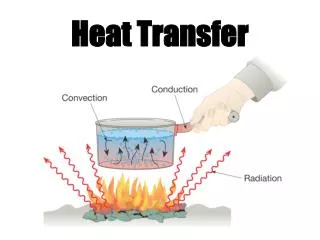
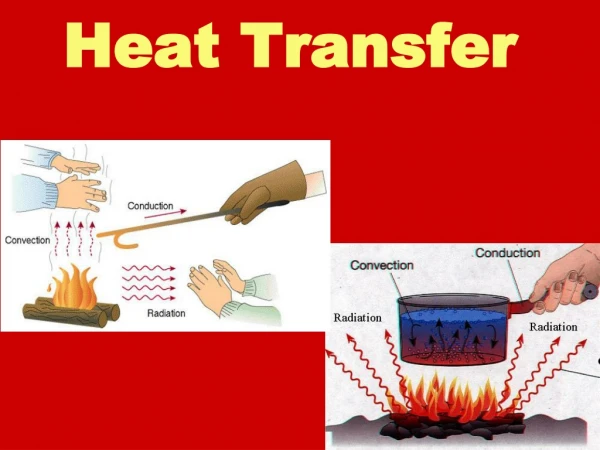
Modes of Heat Transfer • Diffusion(transfer of heat within one medium or from one medium to another medium) • Conduction (diffusion of heat in moving or stationary rigid bodies) • Convection(diffusion of heat in moving deformable bodies) • Radiation(transfer of heat by electromagnetic waves)
Heat Transfer Coefficient • Convective heat transfer is affected by the geometry, surface condition, and fluid properties. • To find h, the detailed flow field must be known. • Mass (1) • Momentum (3) • Energy (1) • Equation of State (1) • Solve for u, v, w, T, P, and ρ • Find qw = h A (TW -T∞) {and also tw =m du/dyw} • Π-Buckingham Theorem (f = n – m) • h = h (k, m, cp, r, L, e, DT, gbDT, hst, t) • Nu = Nu (Gr, Pr, Ste, Fo, e/L)
Boundary Conditions • Prescribed Temperature • Prescribed Heat Flux (Fourier’s Law) • Insulation • Convective (Newton’s Cooling Law) • Radiative (Stefan-Boltzmann Law) • Prescribed Heat Flux Acting at a Distance • Interface (Continuity, Conservation Law) • Moving Boundary
How to insulate a boundary? • q 0 • Efficient electric heater Guard Heater
When is the assumption of isothermal or insulated wall justified? • h 0; h ∞ • Flow through a thin heated tube • Insulated wall [ ] Ti Tm (Boiling) • k 0; k ∞ • Lumped vs. distributed (Biot number) • Insulated wall k large (lumped) k small (distributed) • Bare tube, h0<< hi (qo0)
Moving Boundary • Ice layer forming on surface of a pond on a clear night with calm wind Tsky = 0 K; T∞ = 3oC; Tℓ = 4oC; Tice = 0oC)