Heat Transfer
330 likes | 882 Vues
T 1. T 2. Q. Heat Transfer. In the previous sections, we learned that heat can flow from one object to another (e.g., from the warmer to the cooler object) In this section, we investigate the details of how this heat is transferred

Heat Transfer
E N D
Presentation Transcript
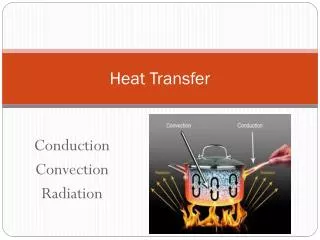
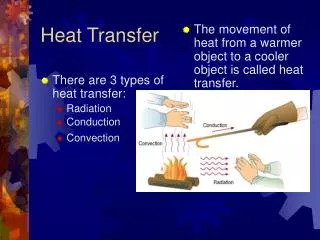
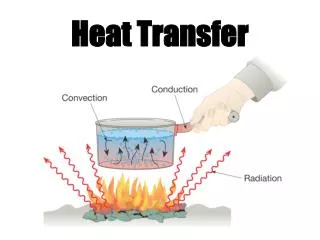
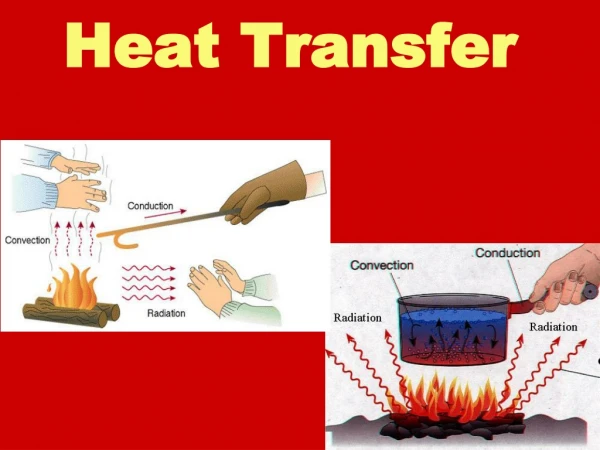
T1 T2 Q Heat Transfer • In the previous sections, we learned that heat can flow from one object to another (e.g., from the warmer to the cooler object) • In this section, we investigate the details of how this heat is transferred • Heat is transferred by three methods - Convection - Conduction - Radiation
Convection • Heat is transferred by the bulk movement of a fluid (liquid or gas) • Consider a liquid (water). We know from thermal expansion, that its volume V increases with temperature. • Given the density =m/V, it will decrease with temperature since the mass is constant. • If we heat water in a pot, we apply the heat at the bottom. For some finite mass of the water, its volume increases and density decreases
The water above is cooler and therefore has a larger density (more mass per unit volume). It sinks while the warmer water rises • As the warmer water rises, it transfers heat to the surrounding cooler water until it is the same temperature as the coldest water T1,1 Q Q T2,2 Q • This water then sinks, because all of the water below it is warmer a Convection Current • Convection currents exist in the ocean (Gulf Stream), the sun and stars, …
This kind of convection is called Natural Convection • Forced Convection – some external device (fan, water pump) establishes the convection currents • Unfortunately, the mathematical description of convection is beyond the scope of this course Conduction • Heat is transferred directly through the material or from one material to another (solids, liquids, and gases) k A T1 Q T2 L T1 T2
The heat conducted is where L=length heat flows along [m] A=cross sectional area [m2] t=time over which heat is transferred [s] k=thermal conductivity of the material, see Table 16-3, has units of [W/(m C°)] or [J/(s m C°)] • Large k – more heat flow – thermal conductor (good electrical conductors are good thermal conductors – metals) • Small k – heat flow is restricted – thermal insulators
Example Problem Three building materials, plasterboard [k1=0.30 J/(s m C°)], brick [k2=0.60 J/(s m C°)], and wood [k3=0.10 J/(s m C°)], are sandwiched together. The temperature at the inside and outside surfaces are 27 °C and 0 °C, respectively. Each material has the same thickness and cross-sectional area. Find the temperature (a) at the plasterboard-brick interface and (b) at the brick-wood interface. Solution: Consider conductive heat flow at each interface Given: A1=A2=A3=A, L1=L2=L3=L
Find T12 and T23 Q1=Q2=Q3 a) T12 T23 Inside,0 Outside,4 1 2 3 T0=T01=27 °C Q1 Q2 T4=T34=0 °C Q3 L L L Two unknowns? Stop here go to part b)
Radiation (Thermal) • Heat is transferred by electromagnetic waves: microwaves, radio waves, infrared radiation, visible (optical), ultraviolet radiation, x-rays, gamma rays • ``Radiation’’ is more general than nuclear radiation (alpha, beta, gamma particles) • Every object emits radiation and absorbs radiation • The temperature, surface area, and surface properties of an object effects the amount of heat that is emitted or absorbed and the wavelength
An object which absorbs 100% of the radiation is known as a blackbody – a perfect absorber. • An object which is a perfect absorber is a perfect emitter. The radiation emitted by a blackbody is called blackbody radiation. • The absorption/emission properties of an object are described by its emissivity, 0<e1 and is unitless. e=1 for a black-body. • The heat due to radiation is where = Stefan-Boltzmann =5.669x10-8 J/(s m2 K4) and T must be in [K]
Example Problem The filament of a light bulb has temperature of 3.0x103°C and radiates 60 W of power. The emissivity of the filament is 0.36. Find the surface area of the filament. Solution: Given: P=Q/t=60 W, e=0.36, T=3000 °C=3273.15 K Remember that 1 W= 1 J/s