Phosphate Ester Formation
Learn about the formation of phosphate esters and amino sugars in carbohydrate metabolism. Explore the role of these compounds in nucleotides, energy carrying molecules, and chemical messengers. Understand the process of glycosidic bond formation and disaccharide formation, as well as the different types of polysaccharides and their properties. Discover the importance of glycolipids and glycoproteins in cell recognition processes. Consider dietary implications and the glycemic index.

Phosphate Ester Formation
E N D
Presentation Transcript
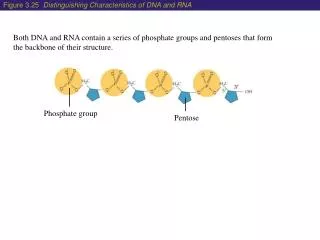
Phosphate Ester Formation • Hydroxyl group + H3PO4 --> Phosphate Ester • Phosphate esters are important compounds in carbohydrate metabolism.
Phosphate ester formation -D-glucose-1-phosphate Sugar and phosphate combinations are the basis for nucleotides involved in DNA / RNA, energy carrying molecules (ADP & ATP), and chemical messengers (cAMP)
Amino Sugar Formation • Hydroxyl group is replaced by an amino group --> Aldosamine • Important in cartilage polysaccharides and red blood cell markers (ABO) • There are 3 important, naturally-occurring amino sugars. • In each the amino group is on C#2.
Amino sugar formation Glucosamine and hyaluronic acid act as the backbone for the formation of proteoglycans found in the structural matrix of joints
Glycosidic bonds: Thehydroxyl groupand ahydroxyl group of another sugar or other compound can join together, splitting out water to form aglycosidic bond. R-OH+HO-R' R-O-R' +H2O Acting as an alcohol Acting hemiacetal glycosidic linkinghelps form disaccharides, oligosaccharides, and polysaccharides from rings of monosaccharides
Disaccharide formation Alpha () or beta () describes the–OH orientationon C #1. The numbers represent theC # connectionson the Haworth projection
Disaccharides • Formation of disaccharides is like glycoside formation (condensation rxn) • Monosaccharide + alcohol --> glycoside + H2O • Monosaccharide + monosaccharide --> disaccharide + H2O • Disaccharideglycosidic linkage Reducing?Human Digestion • Maltose (1-4) yes easily • Cellobiose (1-4) yes no • Lactose (1-4) yes usually • Sucrose (1-2) no yes
Common Disaccharides: Sucrose: a-Glucose and b-Fructose a, b (12) glycosidic linkage Milk sugar: galactose and glucose connected a(14) glycosidic linkage of 2 D-Glucose molecules
Polysaccharides (glycan) • Variations • Homopolysaccharides vs. Heteropolysaccharides • Length of chain • Type of Glycoside Linkage • Degree of Branching • Properties • NOT sweet • No positive Tollens or Fehling’s test • Limited water solubility • Colloids form readily
Starch (plants) amylose (15-20%) straight-chain -glucose polymer (~1000 G) amylopectin (80-85%) branched chain (~100,000 G) glucose polymer (~100,0000 G) Starch + H2O --> glucose Nutritional value Glycogen (animals) Highly branched glucose polymer (~1,000,000 G) Glucose <==> Glycogen (stored in liver & muscle) Storage Polysaccharidesenergy source (homopolysaccharides)
Cellulose (cell wall) straight chain -glucose polymer Chitin (exoskeleton) N-acetyl amino derivative of glucose Structural Polysaccharides(homopolysaccharides)
Cotton and wood are primarily cellulose (14) glycosidic bonds of two glucose rings create linear but angled bonding. Our enzymes cannot match this bond angle structure to hydrolyze cellulose into cellobiose subunits or break that down to glucose
Hyaluronic acid Joint lubricant Heparin Anticoagulant Acidic Polysaccharides(heteropolysaccharides) Hyaluronic acid: heteropolysaccharide – 2 different glucose derivatives: Glucuronic acidplus N-acetyl-b-D-Glucosamine Alternates b(13) and b(14) linkage Acidic polysaccharides associated with the connective tissue of joints give hurdlers such as these the flexibility needed to accomplish their task.
Glycolipids & Glycoproteins • These form when glycosidic linkages connect monosaccharides with lipids &/or proteins. • Very important molecules for cell recognition processes
Dietary Considerations • A balanced diet ~ 60% carbohydrate • Simple carbs = mono & disaccharides • Complex carbs = polysaccharides • Starch • Cellulose • Natural vs. Refined Sugars • Natural: a mixture of sugar and other compounds • Refined: 100% sugar molecules • Glycemic effect: due to the rate of carbohydrate digestion • Glycemic index
Glycemic Index: Measures the rate that specific carbohydrates are hydrolyzed into glucose. Slow release of glucose into blood = good Quick release of glucose into blood / overproduction of insulin = bad