Energy Flows
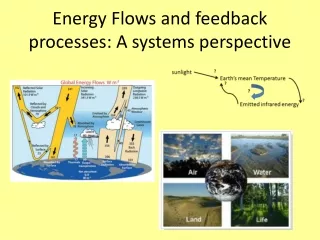
Energy Flows. Biogeochemical Cycles. Earth as a closed system. Energy like sunlight can enter the earth, but matter does not enter or leave. (unless cosmic particles enter the earth) Each of the elements that is vital for life exists on Earth in a closed loop of cyclical changes

Energy Flows
E N D
Presentation Transcript
Energy Flows Biogeochemical Cycles
Earth as a closed system • Energy like sunlight can enter the earth, but matter does not enter or leave. (unless cosmic particles enter the earth) • Each of the elements that is vital for life exists on Earth in a closed loop of cyclical changes • From a systems point of view, Earth is essentially a closed system with respect to matter. • Our class (when the door is closed) as a closed system or a submarine
Continued • Matter does not leave • Therefore from an environmental perspective all resources are interconnected therefore every decision may positively or negatively affect another system • Sunlight and radiant energy can be lost, light (ex. Of open system)
Energy • Energy is the capacity to do work and is measured in calories • A calorie is the amount of heat necessary to raise one gram or one ml of water one degree celcius starting at 15 degrees • Energy comes in many forms: • Radiant (sun) • Chemical (stored in chemical bonds of molecules) • Heat- mechanical and electrical • Energy differs from matter-does not have mass nor occupy space • But it affects matter by making it do work
Energy Continued • Kinetic energy- caused by an object’s motion and mass • Potential energy- stored for later use • Using a hydro dam as an example: what part of the dam would label kinetic energy and potential energy
Kinetic Energy • Water stored behind a hydro dam is potential energy and then when it flows through the dam and turns the turbine it becomes kinetic energy
The Laws of Thermodynamics • Energy follows rules that are explained through the laws of thermodynamics • First law of Thermodynamics- energy can never be created or destroyed, only changed from one form to another • Ex. Solar energy can be stored in plants which eventually die, decompose and become fossil fuels (takes a long time) • Second Law of Thermodynamics- when energy changes it is converted from a general, more concentrated and useful form to a less general, less concentrated useful form • Ex. Liquid water turning into vapour may eventually lose its heat energy
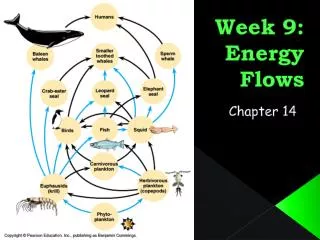
Flow in Ecosystems • All energy used by living things come from the sun and is passed through the food chain to carnivores • At each trophic level energy is lost
Flow of energy in ecosystems continued • All energy used by living things comes from the sun • Through photosynthesis, producers store some of the sun’s energy in foods they make. Ex. lettuce • They use some of this food for their own use and the rest is stored • Herbivores get their energy from eating producers • Carnivores get their energy by eating herbivores or other carnivores
Energy loss in ecosystems • Up to 90% of energy can be lost with each transfer from one trophic level to the next • The more levels there in a food chain more usable energy will be lost • The flow of energy is one way because it cannot be recaptured 100% • Energy flow is one-way and in order for an ecosystem to operate it must always get its energy from the sun
Biogeochemical Cycles • Micro and Macro nutrients are needed the building blocks for living organisms
Biogeochemical Cycles • Nutrients are recycled on earth through interaction between living organisms and the physical environment • Sometimes the interactions are called biogeochemical cycles • Nutrients are constantly circulating through the earths atmosphere, hydrosphere, lithosphere • The 5 main cycles that are driven by incoming solar radiation and gravity are: • Hydrological, carbon, oxygen, nitrogen and phosphorus cycles
Hydrological Cycle-what goes around comes around • Water can be found in liquid, vapour and solid form • 97% of the water found in the hydrosphere is found in the oceans • Of the remaining 3 %, 70% of it is stored in ice • 30% is found under the surface as groundwater • Less than 1% is found in rivers and lakes • Water cycle is the continual circulation of water through the hydrosphere
Hydrological Cycle • Most of the water is found in the oceans • The sun heats the water in the oceans causing it to evaporate in gas form or water vapour • When the water rises up into the atmosphere it cools and condenses around dust and particulate matter • Clouds are formed and eventually the water vapour becomes too heavy and it then falls as rain, snow, sleet, hail, or becomes fog
Hydrological Cycle • Once the water falls on the earth it may flow over the ground into a body of water, this is called runoff • Runoff flows into rivers, streams, creeks, lakes and eventually back into the sea or ocean • Then the whole cycle starts again • Groundwater –water can also be stored in the small spaces found beneath the earth’s surface • An estimated 1170 cubic km of water is cycled like this each day
Hydrological Cycle-Human Impacts • Cloud seeding (silver iodide) • Cloud seeding is the process of spreading either dry ice, or more commonly, silver iodide aerosols, into the upper part of clouds to try to stimulate the precipitation process and form rain. (Beijing Olympics) • Storage of water for human use • Redistribution of water for human use • Building storage containers to control floods • Clearing land for more development
Carbon Cycle-what goes around comes around • In a biogeochemical cycle a chemical or compound changes as it moves through the earth’s system • Carbon which is an element is a building block for life • Carbon can be found in organic materials and minerals like coal and oil • It is not only solid as it can be found in gases such at methane and carbon dioxide
Carbon Cycle- how does it enter the atmosphere? • Carbon can enter the atmosphere by animals breathing out • Organisms that break down decaying matter give off carbon dioxide • When carbon based matter burn, like trees being burned in a forest fore- they release carbon dioxide • Volcanic eruptions can also give off carbon dioxide that was held deep within the earth • Carbon dioxide and also be released or diffused from oceans where c02 is disolved
Carbon Cycle-how is it removed from the atmosphere? • Plants remove carbon dioxide from the atmosphere through photosynthesis and convert it to other compounds like sugars • The process also releases oxygen into the atmosphere • The carbon that is stored in plant tissues in the form of carbohydrates are then passed to animals that eat the plants
Carbon Cycle- how does phytoplankton play a role in this cycle? • Phytoplankton are ocean plants and like land plants they also conduct photosythesis • During photosynthesis the plants convert carbon dioxide into oxygen which is then diffused out of the water • Most phytoplankton is eaten by marine animals • Some phytoplankton decay and fall to the ocean floor • The decaying phytoplankton become part of the ocean sediment and the carbon that was stored in the plant becomes part of the ocean floor • This is why oceans are considered to be a “carbon sink” because it stored a lot of carbon
Carbon Cycle- facts • Oceans remove 40 % of the carbon that is released by the burning of fossil fuels into the atmosphere • Carbon can be stored for long periods of time • Sometimes it can be stored for the lifespan of the plant or animal • When the plant or animal dies the carbon changes in the decomposing process and carbon can be found in the form of methane • If a plant or animal dies in low oxygen environment it can eventually be turned into a fossil fuel, like oil and coal • Carbon like water can never be destroyed, it only changes from one form to another
Carbon Cycle- Human Impacts • Humans have increased the amount of C02 in the atmosphere due to the burning of fossil fuels • Humans have also increased C02 levels by the encroachment of people and development in areas that are rich in vegetation (forests) • Explain the impacts the above statements may have on the environment and how they add C02 to the atmosphere • What would happen to the carbon cycle if we continue to pollute our Oceans?
Nitrogen Cycle-what goes around comes around • Nitrogen is the most abundant element in the atmosphere • The nitrogen cycle is complicated as it converts nitrogen from its inert (not useful) form to one that can be used • A total of 5 steps can be found in the nitrogen cycle where it is converted from a gas to compounds that can be used by plants
Nitrogen Cycle- basics • Nitrogen cycle starts with nitrogen fixation – converts atmospheric nitrogen to ammonia • This conversion is done by nitrogen-fixing bacteria (rhizobia) • Ammonia can also be found in plant and animal tissue • The most important way plants get nitrogen is through dead plant matter mineralizing nitrogen back into the soil • So ammonia is converted from organic matter that is decaying into nitrogen in mineral form
Nitrogen Cycle and Eutrophication-Human Impacts • Eutrophication is a process by which plant growth increases in a lake or pond (sound good?) • In time, eutrophication may cause plants to completely fill in the area where a lake or pond once stood • The word "eutrophic" is of Greek origin, meaning "truly nourished.“
Nitrogen Cycle and Eutrophication- Human Impacts • The accelerated growth and overcrowding of plants is due to either natural fertilizing agents that are washed from the soil, or the runoff of chemical fertilizers applied to agricultural lands (fertilizers have lots of nitrogen in it as it helps plants grow quickly and abundantly) • Eutrophication may also be brought about by the drainage of sewage, industrial wastes, or detergents into a body of water
Nitrogen Cycle and Eutrophication- Human Impacts • As the overcrowded plants die off, the dead and decaying vegetation depletes the lake's oxygen supply (why?) • The bacteria that help to decompose dead plant matter use up a lot of oxygen • This, in turn, leads to the death of fish in the lake. The accumulated dead plant and animal material eventually changes a deep lake to a shallow one. • The shallow lake then becomes a swamp, and finally it becomes dry land
Good or Bad? • With your seat partner create a list of pros and cons of nitrogen eutrophication
Phosphorous Cycle- what goes around comes around • Unlike any other biogeochemical cycle the atmosphere does not play a big role in the phosphorous cycle • Why? Phosphorous are usually solids on earth • The largest reservoir of phosphorous can be found in sedimentary rock
Phosphorous Cycle-how does it work on land? • When rain weathers phosphorous in rocks it is taken to other locations like soil and water • Plants take up the phosphorous from the soil t • Animals eat the plants that contain phosphorous and it is absorbed in the animal tissue • The phosphorous is then returned back to through animal urine, and feces and from decomposition once plants and animals die
Phosphorous Cycle-how does it work in water? • Much like land the phosphorous attach itself to marine plant life • The phosphorous in this case is deposited when soil particles reach the bodies of water from erosion, run-off and settle on the ocean floor or lake bottoms • When sediments are stirred up, phosphates are released • Water plants take up the phosphates and it then enters the aquatic food chain • Phosphorous can move quickly through plants and animals, but it moves slowly through soil or the ocean floor
Phosphorous- Human Impacts • Concentration of large numbers of organisms such as humans, cattle, pigs, creating heavy burdens of phosphate-rich waste materials • How would the above example enter the freshwater systems • Create a flow chart or another graphic organizer to explain the flow. ( concept map, fishbone, diagram) • Explain how too much phosphorous can accelerate eutrophication in fresh water ways?