Stoichiometry Calculations in Chemistry: Mole-Mass Conversion Examples
Learn how to convert moles to mass and vice versa in stoichiometry with practical examples including chemical equations. Calculate reactants needed and products formed.

Stoichiometry Calculations in Chemistry: Mole-Mass Conversion Examples
E N D
Presentation Transcript
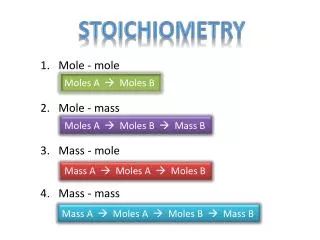
Stoichiometry 1. Mole - mole Moles A Moles B 2. Mole - mass Moles A Moles B Mass B 3. Mass - mole Mass A Moles A Moles B 4. Mass - mass Mass A Moles A Moles B Mass B
A B N2 + H2 3 2 NH3 How many moles of ammonia will be produced from 4.5 moles of hydrogen? mass A moles A moles B mass B 2 mol NH3 4.5 mol H2 x = 3 mol NH3 3 mol H2
B HgO Hg + O2 2 2 A How many moles of mercury (II) oxide are needed to produce 125 g of oxygen? mass A moles A moles B mass B 1 mol O2 2 mol HgO 125 g O2 x x = 7.81 mol HgO 32.0 g O2 1 mol O2
A B Fe3O4 + CO Fe + CO2 4 3 4 What mass of Fe can be produced from the complete reaction of 4.0 moles of CO? mass A moles A moles B mass B 3 mol Fe 55.85 g Fe 4.0 mol CO x x = 4 mol CO 1 mol Fe 167.55 g Fe
Fe3O4 + CO Fe + CO2 4 3 4 A What mass of CO2 can be produced from 25.0 g of Fe3O4? B mass A moles A moles B mass B 1 mol Fe3O4 4 mol CO2 44.01 g CO2 25.0 g Fe3O4 x x x = 231.55 g Fe3O4 1 mol Fe3O4 1 mol CO2 19.01 g CO2
WO3 + H2 W + H2O 3 3 What mass of hydrogen is needed to produce 75.0 g of tungsten?
2 Al + H2SO4 Al2(SO4)3 + H2 3 3 If 37.52 grams of aluminum reacts, what mass of aluminum sulfate will be produced?
Fe + CuSO4 Fe2(SO4)3 + Cu How many moles of CuSO4 is required to produce 132.6 grams of Cu?
AgNO3 + BaCl2 AgCl + Ba(NO3)2 What mass of barium chloride is needed to produce 235.50 grams of silver chloride?