Gene Expression Analysis by SAGE
Gene Expression Analysis by SAGE. Gene Expression. Some challenges: Large number of genes How do you keep samples and equipment small and affordable? How do you analyze that amount of data? Need to know sequence of some or all genes. Methods. SAGE (Serial Analysis of Gene Expression)

Gene Expression Analysis by SAGE
E N D
Presentation Transcript
Gene Expression Some challenges: • Large number of genes • How do you keep samples and equipment small and affordable? • How do you analyze that amount of data? • Need to know sequence of some or all genes
Methods • SAGE (Serial Analysis of Gene Expression) • Victor E. Velculescu, Lin Zhang, Bert Vogelstein, Kenneth W. Kinzler • October 1995
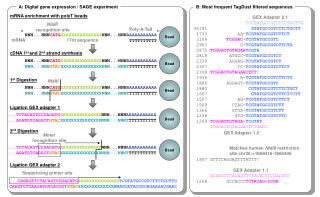
SAGE – introduction • SAGE uses short tags of cDNA made from all mRNAs in a cell • The assumption: a tag of only 9 or 10 bases, from a specific position in the mRNA, is enough to identify the transcript • These tags are linked together so that they can be cloned in large groups • Sequencing identifies genes, and they can be counted to determine relative expression
SAGE – the method The 3’ end of biotinylated, double stranded cDNA adheres to streptavidin beads This and all other pictures of the SAGE method are from http://sciencepark.mdanderson.org/ggeg/SAGE_technique.htm
SAGE – the method • Anchoring enzyme (AE) usually recognizes a 4-bp site • Restriction may occur before binding to beads • cDNA is divided in half, and each half is ligated to a different linker (A or B) • Linkers have a IIS restriction enzyme site and are complementary to a PCR primer
SAGE – the method • Tagging enzyme is a type IIS restriction enzyme • Unique in that they cleave at a defined distance away from the site that they recognize • Tagging enzyme recognizes site in the linker, and cuts off a short “tag” of cDNA, leaving a blunt end. • Two groups of cDNAs are ligated to each other, to create a “ditag” with linkers on either end
SAGE – the method • PCR is used to amplify the ditags, using a primer that is complementary to the linker. • The cDNA is again digested by the AE, breaking the linker off right where it was added in the beginning. This leaves a “sticky” end with the sequence GTAC (or CATG on the other strand) at each end of the ditag.
SAGE – the method • Ditags are ligated together to form long concatemers. Between each ditag is the AE site, allowing the scientist and the computer to recognize where one ends and the next begins. • The concatemers are sequenced, and the tags are matched up with the gene that they uniquely represent. By counting the number of times each tag appears, the relative expression levels can be determined.
SAGE – advantages • No hybridizing, so no cross-hybridizing can occur • Knowledge of genes to be detected can wait until after experiment • Can help identify new genes by using tag as a PCR primer
SAGE – disadvantages • Cost and time required to perform so many PCR and sequencing reactions • Type IIS restriction enzyme can yield fragments of the wrong length depending on temperature. • Multiple genes could have the same tag • As with microarrays, mRNA levels may not represent protein levels in a cell
MOUSE SAGE SITE http://mouse.biomed.cas.cz/sage/compare.cgi