Understanding Molar Conversions: The Size and Mass of a Mole Explained
Explore the concept of the mole, a unique counting unit in chemistry comparable to a dozen. Learn about Avogadro’s number (6.02 x 10^23), which quantifies the number of entities in one mole. Discover fascinating examples that illustrate the incredible scale of a mole, from pennies covering the Earth to hockey pucks equaling the Moon's mass. Understand molar mass calculations, with examples including water, sodium chloride, and more. Unlock the secrets of moles, conversions, and practical applications in chemistry.

Understanding Molar Conversions: The Size and Mass of a Mole Explained
E N D
Presentation Transcript
The Mole Molar Conversions
VERY A large amount!!!! A. What is the Mole? • A counting number (like a dozen) • Avogadro’s number (NA) • 1 mol = 6.02 1023 items
This is the fundamental definition of what one mole is: • One mole contains as many “entities” (things) as there are atoms in 12 grams of carbon-12. • One mole of carbon-12 contains 6.02 x 1023 atoms of carbon-12.
A. What is the Mole? HOW LARGE IS IT??? • 1 mole of pennies would cover the Earth 1/4 mile deep! • 1 mole of hockey pucks would equal the mass of the moon! • 1 mole of basketballs would fill a bag the size of the earth!
B. Molar Mass • Mass of 1 mole of an element or compound. • Atomic mass tells the... • atomic mass units per atom (amu) • grams per mole (g/mol)
B. Molar Mass Examples 12.01 g/mol 26.98 g/mol 65.39 g/mol • carbon • aluminum • zinc
B. Molar Mass Examples • water • sodium chloride • H2O • 2(1.01) + 16.00 = 18.02 g/mol • NaCl • 22.99 + 35.45 = 58.44 g/mol
B. Molar Mass Examples • sodium bicarbonate • sucrose • NaHCO3 • 22.99 + 1.01 + 12.01 + 3(16.00) = 84.01 g/mol • C12H22O11 • 12(12.01) + 22(1.01) + 11(16.00) = 342.34 g/mol
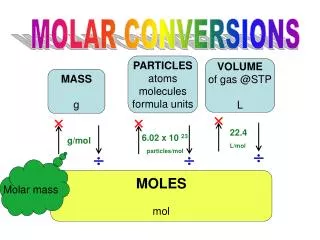
MASS IN GRAMS MOLES NUMBER OF PARTICLES C. Molar Conversions molar mass 6.02 1023 (g/mol) (particles/mol)
C. Molar Conversion Examples • How many moles of carbon are in 26 g of carbon? 26 g C 1 mol C 12.01 g C = 2.2 mol C
C. Molar Conversion Examples • How many moles of sodium chloride are in 26 g of sodium chloride? 26 g NaCl 1 mol NaCl 58.44 g NaCl = 0.44 mol NaCl
C. Molar Conversion Examples • How many moles of magnesium fluoride are in 85 g of MgF2? 85 g MgF2 1 mol MgF2 62.31 g MgF2 = 1.36 mol MgF2
MASS IN GRAMS MOLES NUMBER OF PARTICLES C. Molar Conversions molar mass 6.02 1023 (g/mol) (particles/mol)
C. Molar Conversion Examples • How many molecules are in 2.50 moles of C12H22O11? 6.02 1023 molecules 1 mol 2.50 mol = 1.51 1024 molecules C12H22O11
C. Molar Conversion Examples • How many molecules are in 2.50 moles of C2H6? 6.02 1023 molecules 1 mol 2.50 mol = 1.51 1024 molecules C2H6
C. Molar Conversion Examples • Find the mass of 2.1 1024 molecules of NaHCO3. 2.1 1024 molecules 1 mol 6.02 1023 molecules 84.01 g 1 mol = 290 g NaHCO3
C. Molar Conversion Examples • How many atoms are in 10 grams of NaCl. 1 mol 58.44 g NaCl 6.021023 atoms NaCl 1 mol 10 g NaCl = 1.03x 1023 atoms NaCl
MOLE DAY FUNNIES • Q: What did Avogadro teach his students in math class? • Moletiplication
Q: What kind of fruit did Avogadro eat in the summer? • A: Watermolens
Why was there only one Avogadro? • When they made him, they broke the Moled
What did Avogadro invent for his wife to use as a night cream? Oil of Molay