2 pt
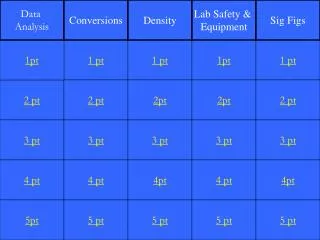
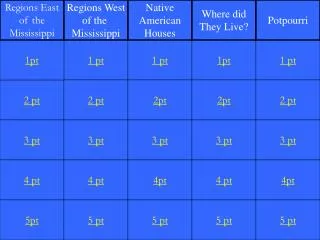
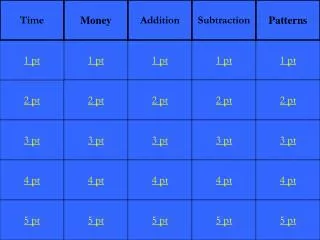
Data Analysis. Conversions. Density. Lab Safety & Equipment. Sig Figs. 1pt. 1 pt. 1 pt. 1pt. 1 pt. 2 pt. 2 pt. 2pt. 2pt. 2 pt. 3 pt. 3 pt. 3 pt. 3 pt. 3 pt. 4 pt. 4 pt. 4pt. 4 pt. 4pt. 5pt. 5 pt. 5 pt. 5 pt. 5 pt. Please select your Team. Team 1 Team 2 Team 3

2 pt
E N D
Presentation Transcript
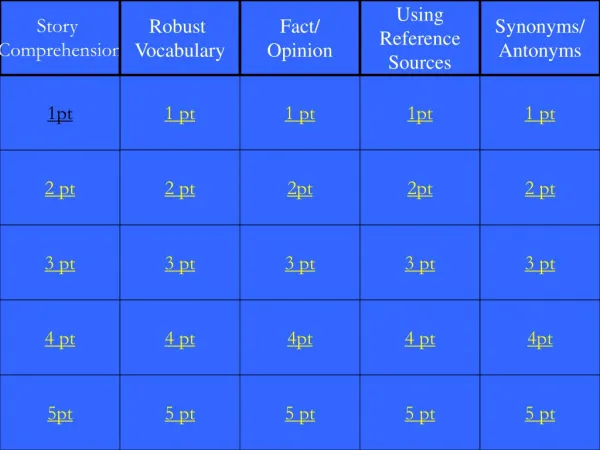
Data Analysis Conversions Density Lab Safety & Equipment Sig Figs 1pt 1 pt 1 pt 1pt 1 pt 2 pt 2 pt 2pt 2pt 2 pt 3 pt 3 pt 3 pt 3 pt 3 pt 4 pt 4 pt 4pt 4 pt 4pt 5pt 5 pt 5 pt 5 pt 5 pt
Please select your Team. • Team 1 • Team 2 • Team 3 • Team 4 • Team 5 • Team 6
Data Analysis1 point question • When a test instrument is calibrated, does its accuracy, precision, or reliability improve?
10 Please make your selection... • precision • accuracy • reliability • All of the above Scores
Data Analysis 2 point question • Which of the following measurements (of different masses) is the most accurate?
10 Please make your selection... • 3.1000 g • 3.10000 g • 3.12222 g • 3.000000 g Scores
Data Analysis 3 point question • Three different people weigh a standard mass of 2.00 g on the same balance. Each person obtains a reading of 7.32 g for the mass of the standard. These results imply that the balance that was used is____
10 Please make your selection... • accurate • precise • accurate and precise • neither accurate nor precise Scores
Data Analysis 4 point question • In the measurement 0.503 L, which digit is the estimated digit?
10 Please make your selection... • 5 • the 0 immediately to the left of the 3 • 3 • the 0 to the left of the decimal point Scores
Data Analysis 5 point question • The technique of dimension analysis is used to
10 Please make your selection... • Convert a very large unit into an easier-to-use form. • Determine the independent variable on a graph • Convert units of measurement • Develop a testable hypothesis Scores
Conversions1 point question • How many meters are in a kilometer?
10 Please make your selection... • 1 • 10 • 100 • 1000 Scores
Conversions2 point question • An intramuscular medication is given at 5.00 mg/kg of body weight. If you give 425 mg of medication to a patient, what is the patient’s weight in pounds?
10 Please make your selection... • 187 • 190 • 200 • 208 Scores
Conversions3 point question • How many fl. oz. are equivalent to 4 liters in volume?
10 Please make your selection... • 73 • 125 • 135 • 100 Scores
Conversions4 point question • Consider a fixed distance, like the diameter of the earth. In which of the following units would the number expressing this distance be the largest?
10 Please make your selection... • miles • kilometers • feet • meters Scores
Conversions5 point question • How many oz are found in 245.21 hm3?
10 Please make your selection... • 1.1709x10-5 • 1.1710x10-5 • 1.1709x1013 • 1.1710x1013 Scores
Density1 point question What is more dense a ton of aluminum or a ton of iron?
10 Please make your selection... • A ton of aluminum • A ton of iron • Both are equally dense since they have the same weight. • Can not be determined Scores
Density2 point question • What is the volume of 80.0 g of ether if the density is 0.70 g/ml?
10 Please make your selection... • 5.6 x 101 • 8.8 x 10-3 • 1.1 x 102 • 8.0 x 101 Scores
Density3 point question • What is the density of an object having a mass of 8.0 g and a volume of 25 cm3?
10 Please make your selection... • 0.32 g/cm3 • 2.0 g/cm3 • 3.1 g/cm3 • 200 g/cm3 Scores
Density4 point question • An object has a mass of 3.15 oz. When it is submerged in a graduated cylinder containing 325.2 ml of water, the water level rises to 442.5 ml. What is the density (g/ml) of the object?
10 Please make your selection... • 1.23 g/ml • 3.35 g/ml • 0.987 g/ml • 0.762 g/ml Scores
Density5 point question • If the temperature of a piece of steel decreases, what happens to its density?
10 Please make your selection... • The density decreases • The density increases • The density does not change. • The density first increases, then decreases Scores
Lab Safety and Equipment1 point question • What is the proper name of the lab equipment shown in the picture to the left?
10 Please make your selection... • plastic mesh • metal square • wire gauze • strainer Scores
Lab Safety and Equipment2 point question • In the following diagram, a meter stick is being used to measure the length of an object. What is the correctly measured length?
10 Please make your selection... • 0.280m • 0.28m • 0.2m • 2.8m Scores
Lab Safety and Equipment3 point question • When working with volatile chemicals, where should experiments be conducted?
10 Please make your selection... • While wearing a face shield. • Inside a fume hood. • In a room with the window open. • In a temperature controlled room. Scores
Lab Safety and Equipment4 point question • What is the proper name of the lab equipment shown in the picture to the left?
10 Please make your selection... • watch glass • glass bowl • mortar • beaker Scores
Lab Safety and Equipment5 point question • When diluting an acid, you should do which of the following?
10 Please make your selection... • Mix the water into the acid. • Evaporate water from the acid using heat. • It is not possible to dilute acids. • Mix the acid into the water. Scores
Sig Figs1 point question • How many significant figures are in the following number? • 3,000,000
10 Please make your selection... • 0 • 1 • 4 • 7 Scores
Variables 2 point question • How many significant figures are in the following number? • 0.000340023
10 Please make your selection... • 4 • 6 • 10 • 0 Scores
Variables 3 point question • Solve the following problem taking significant figures into account: • 16.5 + 8 + 4.37
10 Please make your selection... • 28.9 • 28.87 • 29 • 30 Scores
Variables 4 point question • Solve the following problem taking significant figures into account: • 3.15 x 2.5 x 4.00
10 Please make your selection... • 32 • 31.5 • 30 • 31.50 Scores