Western Blotting
Western Blotting. The experiment mainly focuses on the steps involved in transferring the separated samples from gel to the membrane and detect the desired protein by using the fluorescent/enzyme tagged antibodies. Related Los: Transfer of gel to membrane, fluorescent/enzyme tagged antibodies

Western Blotting
E N D
Presentation Transcript
Western Blotting The experiment mainly focuses on the steps involved in transferring the separated samples from gel to the membrane and detect the desired protein by using the fluorescent/enzyme tagged antibodies. • Related Los: Transfer of gel to membrane, fluorescent/enzyme tagged antibodies > Prior Viewing- IDD-11. Protein quantification, IDD-17. SDS-PAGE > Future Viewing- IDD-25. SDS-PAGE gel analysis, IDD-28. In solution digestion, IDD-30. Matrix Instrumentation • Course Name:Western blotting • Level(UG/PG):UG • Author(s):Dinesh, Vinayak • Mentor: Dr. Sanjeeva Shrivastava *The contents in this ppt are licensed under Creative Commons Attribution-NonCommercial-ShareAlike 2.5 India license
Learning objectives 1 • After interacting with this Learning Object, the learner will be able to: • Define the electro-blotting principle • Operate the steps involved in staining process • Interpret to detect the protein of interest by antibody tagging • Assess the troubleshooting steps involved in the experiments. 2 3 4 5
Master Layout 1 Reagents for western blotting (Slide: 5-17) 2 Semi dry Transfer (Slide: 18) Western blotting (Slide: 19-41) 3 Primary antibody treatment (Slide: 42-46) 4 Secondary antibody treatment(Slide: 47-52) 5
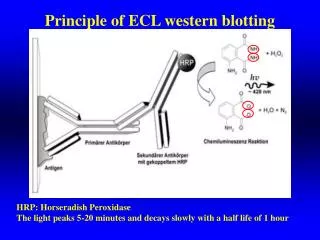
Definitions and Keywords 1 1. Western Blotting: The technique used to transfer the proteins in the gel to the blotting membrane so that presence of desired protein spot can be detected using enzyme conjugated antibodies. 2. Skim milk: Milk devoid of cream or fat is called skim milk which is used for blocking the membrane to prevent the non specific binding of the antibodies. 3. Primary Antibody: Antibody which is specific for the protein of interest. Which has the binding sites for protein of interest and also for secondary antibody. 4. Secondary antibody: Antibody specific for the Fc region of the primary antibody which is also tagged with enzymes like alkaline phosphatase, which gives a color reaction after reacting with the substrate like NBT/BCIP. 5. 5-Bromo-4-chloro-3-indolyl phosphate (BCIP) is a chemical compound used in immunoblotting, with nitro blue tetrazolium (NBT), for sensitive colorimetric detection of alkaline phosphatase. 2 3 4 5
Step 2: T1:Reagents for western blotting 1 2 3 Audio Narration (if any) Description of the action Show a measuring balance, with display, ON, OFF and TARE/0 buttons on it. let user ON it, display reading as 0.000g, let user picks up the paper from the rack, makes 1/10 of folding on the sides and places it on the balance. Now the display reading changes to 0.003g. Instruct user to TARE the reading. And animate to click the tare button. Once user clicks it, reading must show ”0” When measuing with paper, the weight of the paper need to be tared from actual reading. 4 5 Reagents are similar to IDD-32. Buffer preparation for western analysis from slide: 5-17.
Step 3: T1:Reagents for western blotting 1 Tris base Glycine 2 methanol Description of the action Audio Narration 3 Let user pick up glycine, tris base, methanol, spatula, measuring cylinder from the rack and keeps it on the table next to balance. Instruct user to weigh 28.8g of glycine, let user tare the balance, user should click on the glycine bottle, uncap it, with help of spatula weigh the required amount on a paper over the balance. Display a gradual increase in reading with quantity addition. if the gram exceeds user should remove some quantity or if it less add the quantity to get the exact required amount. After weighing transfer the quantity to beaker. Now 4.6g accordingly for tris base. Prepare transfer buffer containing glycine, Tris Base and methanol. Buffer provides wet transfer condition during electro-blotting of proteins from gel to membrane. This wet transfer is recommended for large proteins and avoids drying of the membrane. 4 5
Step 3: T1:Reagents for western blotting 1 Tris base Glycine 2 methanol Description of the action Audio Narration 3 Now to the beaker, take methanol bottle, open the cap, take 1000ml measuring cylinder, measure 900ml. Let user remove the excess methanol if level crosses 900ml mark. Tranfer it to beaker. Now take the beaker, shake it to make a proper mix. Animate the powder getting into the solution. Now tranfer the beaker solution to 1000ml measuring cylinder to makeup the volume to 1000ml by adding methanol. If your are using nitrocellulose, buffer must be prepared using methanol. if using PVDF, distilled water can be used to prepare the buffer. Methanol treatment is needed at the time of electroblotting to activate the PVDF. Now to the beaker, add methanol 4 5
Step 4: T1:Reagents for western blotting 1 Tween 20 Tris base Nacl 2 Kcl Description of the action Audio Narration Let user takes out Nacl, Kcl,Tris Base, tween 20 from the rack and keep it next to balance. Instruct user to weigh 8g of Nacl, 0.2g of Kcl, 3g of tris base and 600ul of tween 20. let user pick the bottle, uncap it, weigh the required amount with help of spatula on a paper over the balance. Display a gradual increase in reading with quantity addition. if the gram exceeds user should remove some quantity or if it less add to get the required amount. After weighing transfer the quantity to beaker. Now take out 1ml pippette, set it for 600ul, take out tween20 bottle, uncap it, pipette and transfer 600ul into the beaker. . 3 Prepare TBST buffer which can be used as destain solution and also for washing the membrane. 4 5
Step 4: T1:Reagents for western blotting 1 Tween 20 Tris base Nacl 2 Kcl Description of the action Audio Narration 3 The pH of the TBST buffer need to set to 7.6. Now to the beaker, take methanol bottle, open the cap, take 1000ml measuring cylinder, measure 900ml. Let user remove the excess methanol if level crosses 900ml mark. Transfer it to beaker. Now take the beaker, shake it to make a proper mix. Animate the powder getting into the solution. Now set the pH to 7.6 by using pH meter. 4 5
Step 5: T1:Reagents for western blotting 1 2 STD 1 STD 2 3 Audio Narration Description of the action Before the pH reading, pH instrument need to be calibrated with standards. Once with STD 1 at pH 4 and with STD 2 at pH 9. Display standard pH bottles and pH instrument placed on a table. Instruct user to caliberate the instrument. Let user ON the instrument. Initially for the pH rod is dipped in water, when user clicks on read button, display must show a reading “7”. now take out the rod rinse it with deionized-water, let user cleans the rod with tissue. Now pick the STD 1 , uncap it, dip the cleaned rod into the solution, user must click read button with display showing “4”. now clean the rod and repeat the step to note down the reading for STD 2. 4 5
Step 5: T1:Reagents for western blotting 1 2 NaOH HCl 3 Audio Narration Description of the action Instruct user to set the pH for TBST pH at 7.6. Now take the TBST bottle, uncap it, dip the cleaned pH rod into the solution. User need to click on read button. Initially display must show a reading 6. now instruct user to add NaOH to adjust the pH. Now allow the user to click on NaOH bottle so that drops of NaOH should be added with filler, user need to mix the solution with glass rod, click on read button and the reading should anywhere near 6.1- 6.3. let user keeps adding the NaOH drop till the desired pH is obtained. and later transfer the beaker solution to 1000ml measuring cylinder to makeup the volume to 1000ml by adding methanol Set the pH of TBST buffer to 7.6. 4 5
Step 6: T1:Reagents for western blotting 1 Tris base Nacl 2 Mgcl2 Description of the action Audio Narration 3 Show the bottles labeled as Nacl, Mgcl2,Tris Base. The user should click on the required reagent bottle and spatula for weighing. Instruct user to weigh 0.585g of Nacl and 0.102 g of Mgcl2 and 1.11g of Tris and, let user pick the bottle, uncap it, with help of spatula weigh the required amount on a paper over the balance. if the gram exceeds he should remove some quantity or if it low add to get required amount. Dissolve the weighed amount by adding 100ml of methanol (instruct user to measure methanol in the measuring cylinder) and giving a brief spin to dissolve it. Prepare Alkaline phosphatase buffer containing Nacl,Mgcl2 and tris base which helps in visualization of bands. 4 5
Step 7: T1:Reagents for western blotting 1 2 NaOH 3 HCl Audio Narration Description of the action Then the beaker containing(labeled as “ALP pH 9.8”) has to be taken near pH meter and allow the user to dip ph rod in the solution. Animate like the user switching on the pH meter. The meter should show pH 8 in the display and instruct user to add NaOH. Now allow the user to click on NaOH so that drops of NaOH should be added in fillers and the reading should increase like.8.1,8.3 and then 8.8,9.2,9.3 and 9..8(desired pH). (follow the instruction like in slide:10 & 11) Adjust the pH of the alakaline phosphatase buffer to 9.8 4 5
Audio Narration Description of the action Step 8: T1:Reagents for western blotting 1 2 1000 500 250 3 100 The animator should draw graduated measuring cylinder as shown in slide with graduation 100ml, 250 ml,500ml,1000ml. The user should click on the appropriate cylinder for usage 4 5
Description of the action/ interactivity Audio Narration (if any) Show the bottles labeled as ethanol, glacial acetic acid, measuring cylinder and water. Instruct user to measure the reagents, amount must be displayed, click on bottle, pick each bottle to pour the required amount in measuring cylinder. In case if the level is more, instruct user to remove the extra solution. Accordingly, animate pouring and increase in the level of solution simultaneously in the measuring cylinder. Later transfer the solution into the fixing solution bottle. Step 9: T1:Reagents for western blotting 1 FIXING SOLUTION 2 WATER 3 Prepare fixing solution by measuring 10ml of methanol, 7 ml glacial acetic acid and make up the volume to 100ml with deionized water. 4 5
Step 10: T1:Reagents for western blotting 1 Sodium Acetate 2 Description of the action Audio Narration 3 Show the bottles labeled as Sodium acetate and acetonitrile . The user should click on the required reagent bottle and spatula for weighing. Instruct user to weigh 8.2g of Sodium acetate and, let user pick the bottle, uncap it, with help of spatula weigh the required amount on a paper over the balance. if the gram exceeds he should remove some quantity or if it low add to get required amount. Dissolve the weighed amount by adding 80ml of water and 20 ml (instruct user to measure methanol in the measuring cylinder) and giving a brief spin to dissolve it. Prepare destaining solution containing sodium acetate and acetonitrile 4 5
Step 11: T1:Reagents for western blotting 1 2 NaOH 3 HCl Audio Narration Description of the action Then the beaker containing(labeled as “Destaining solution pH 4”) has to be taken near pH meter and allow the user to dip ph rod in the solution. Animate like the user switching on the pH meter. The meter should show pH 5 in the display and instruct user to add Hcl. Now allow the user to click on Hcl so that drops of Hcl should be added in fillers and the reading should decrease like 4.8,4.6,4.4,4.2,4) Adjust the pH of the Destaining solution to 4 4 5
Description of the action Audio Narration Step 1: T2:Semi dry Transfer 1 Cathode 2 3 Anode Semi dry transfer is used for the transfer of protein spots from gel onto the membrane. Animator should draw a instrument as shown in the figure. Include the options like start, time and stop. Label as “Semi dry transfer apparatus” 4 5
Description of the action/ interactivity Audio Narration (if any) Step 12: T3:western blotting 1 2 Anode plate of semi dry transfer instrument 3 4 Place the anode side of the semi dry transfer on the table Animator should draw a instrument as in fig and include the sign 5
Step 13: T3:western blotting 1 2 Blotting papers 3 4 5
Description of the action Audio Narration Step 13: T3:western blotting 1 Animator should draw some white sheets in rectangular form. Instruct the user to take the transfer buffer and pour it in the box as given in figure of previous slide. Animate like the user taking the white sheets and place it in the buffer. Instruct the user to make movements on the box like see-saw. Place the blot papers in the transfer buffer and allow it to equilibrate by shaking the box. 2 3 4 5
Step 14: T3:western blotting 1 2 3 Blot paper 4 Transfer plate 5
Description of the action Audio Narration Step 14: T3:western blotting 1 Place the equilibrated blot paper on the semi dry transfer plate Animate like taking the white papers from the transfer buffer and place it on the “anode plate of the semi dry transfer as shown in slide 22 2 3 4 5
Step 15: T3:western blotting 1 2 Poly vinylidene fluoride 3 4 Methanol 5
Step 15: T3:western blotting 1 2 Poly vinylidene fluoride 3 4 Transfer buffer 5
Description of the action Audio Narration Step 15: T3:western blotting 1 Animator should draw a white sheet and in between the two blue sheets as shown in figure slide:24. Instruct the user to click on it to get the name as given in slide The animator should instruct the user to take out the white sheet from the blue covering sheets using the forceps as given in slide and put it in the box containing methanol Run a clock for 1 minute and animate like the taking the white sheet and putting it in the box containing transfer buffer as in slide:24-25 and again show a clock running a minute. 2 Pre-treat the polyvinylidine fluoride membrane in methanol followed by transfer buffer for a minute each. 3 4 5
Step 16: T3:western blotting 1 2 PVDF membrane 3 4 Blotting paper 5
Description of the action Audio Narration Step 16: T3:western blotting 1 Animator should instruct the user to take the PVDF membrane (white sheet) from the transfer buffer using forceps and place it on the “blot papers” as shown in the figure slide 27 Place the PVDF membrane on the blotting paper and ensure that there is no bubble between the membrane and the paper. 2 3 4 5
Step 17: T3:western blotting 1 2 3 Gel 4 5
Step 18: T3:western blotting 1 2 3 gel 4 5
Description of the action Audio Narration Step 18: T3:western blotting 1 2 Place the SDS-PAGE gel on the PVDF membrane and ensure that there is no air bubbles between membrane and the gel. Animate at a glance the IDD-17. SDS-PAGE for user to feel the gel run. Animate like placing the gel on the PVDF membrane and instruct the user to level it. 3 4 5
Step 19: T3:western blotting 1 2 Blotting papers 3 4 5
Step 20: T3:western blotting 1 2 3 4 5
Description of the action Audio Narration Step20: T3:western blotting 1 Instruct the user to take the transfer buffer and pour it in the box as given in figure Instruct and animate like the user taking the white sheets and place it in the buffer Instruct the user to move the box like see-saw. Instruct the user to place the papers on the top of the gel Place the blot papers in the transfer buffer and allow it to equilibrate by shaking the box. Place it on the top of the gel. 2 3 4 5
Step 21: T3:western blotting 1 2 3 4 5
Step 22: T3:western blotting 1 2 3 4 5
Step 23: T3:western blotting 1 2 3 4 5
Description of the action Audio Narration Step 22 &23: T3:western blotting 1 2 Pour the transfer buffer on the blot paper Place the cathode plate on the anode plate containing blotting set up and connect it to the power supply Set current depends on the area of the gel*0.8 factor. Animate like the user pouring the drops of transfer buffer on the top of the blot paper using filler as shown in the slide no 50 Show a cathode plate as in figure slide 36 and instruct the user to place the plate on the blot paper Animate like connecting to the power supply as shown in slide:37 Show a power pack with the options” set current, time” Instruct the user to set the current as 50.4 A, time 1:30 hr. 3 4 5
Step 24: T3:western blotting 1 PVDF 2 Proteins 3 Gel 4 5
Description of the action Audio Narration Step 25: T3:western blotting 1 Animate like the blue squares, red circles, green triangles from the gel is moving downwards to the membrane Animate like the blue, red, green show like disappearing in the gel and appearing on the PVDF membrane as the time progresses. During the transfer the negatively charged proteins migrate from the gel to the membrane in the positive side 2 3 4 5
Description of the action Audio Narration Step 25: T3:western blotting 1 Once the transfer is over remove the membrane using the forceps avoid body contacts. Animator should instruct the user to switch off the power supply after 1:30 hr Instruct the user to remove the plate at the top, blot papers using the forceps to remove the membrane. 2 3 4 5
Audio Narration Description of the action Step 26: T4: Primary antibody treatment 1 2 milk Rocker tray Pour the 5% skim milk solution into the tray with user interaction. Instruct user to stop the rocker, pick the tray containing gel from the rocker, place it on table. Now transfer the membrane into the tray containing milk solution solution. Now animate user control to take the tray containing membrane to be placed on ROCKER. Allow user to set the parameters for the rocker and to start the instrument. Animate see-saw movement for the rocker along with solution movement in the tray 3 Place the membrane in the 5% skim milk solution and keep it on the rocker to block the non-specific sites in the membrane. 4 5
Step 27: T4: Primary antibody treatment 1 P.Antibody 2 diluted TBST tray 3 4 Rocker 5
Description of the action Audio Narration Step 27: T4: Primary antibody treatment 1 Dilute the primary antibody with 1:1000 concentration using TBST and add the antibodies to the membrane and shake gently for 2 hours. Animator should draw a tube labeled as primary antibody and a bottle labeled as TBST. Instruct the user to take 1ul of the antibody and 999ul of TBST using the pipette and transfer it to the new tube(label as “diluted”). Draw a tray as given in the slide 43 containing the membrane Instruct the user to add the antibody from diluted tube to the tray Instruct the user to place it in the rocker and switch on, it should move slowly like see-saw and show a clock running 2 hours 2 3 4 5
Description of the action Audio Narration Step 28: T4: Primary antibody treatment 1 2 TBST 3 Instruct the user to pour some TBST to the tray and keep it in the shaker and show a clock running 10 minutes After 10 minutes show like removing the TBST from the tray and pouring the fresh one (repeat the step 3 times) 4 Wash the membrane with TBST to remove the unbound primary antibodies 5
Audio Narration Description of the action Step 29: T4: Primary antibody treatment 1 2 milk Rocker tray Pour the 5% skim milk solution into the tray with user interaction. Instruct user to stop the rocker, pick the tray containing gel from the rocker, place it on table. Now transfer the membrane into the tray containing milk solution solution. Now animate user control to take the tray containing membrane to be placed on ROCKER. Allow user to set the parameters for the rocker and to start the instrument. Animate see-saw movement for the rocker along with solution movement in the tray 3 Place the membrane in the 5% skim milk solution and keep it on the rocker to block the non-specific sites in the membrane and to prevent the background. 4 5
Step 30: T5: Secondary antibody treatment 1 S.Antibody 2 diluted TBST Alkaline phosphatase buffer 3 Rocker 4 diluted tray 5
Description of the action Audio Narration Step 30: T5: Secondary antibody treatment 1 Dilute the Secondary antibody conjugated with alkaline phosphatase with 1:5000 conc using Alkaline phosphatase buffer and 5ml of TBST and add the antibodies to the membrane Animator should draw a tube labeled as Secondary antibody and a bottle labeled as TBST . Instruct the user to take 1ul of the antibody and 4999ul of ALP and 5ml of TBST using the pipette and transfer it to the new tube(label as “diluted”). Draw a tray as given in the slide 47 containing the membrane Instruct the user to add the antibody from diluted tube to the tray Instruct the user to place it in the rocker and switch on, it should move slowly like see-saw 2 3 4 5
Description of the action Audio Narration Step 31: T5: Secondary antibody treatment 1 2 TBST 3 Instruct the user to pour some TBST to the tray and keep it in the shaker and show a clock running 10 minutes After 10 minutes show like removing the TBST from the tray and pouring the fresh one (repeat the step 3 times) 4 Wash the membrane with TBST to remove the unbound Secondary antibodies 5
Description of the action Audio Narration Step 32: T5: Secondary antibody treatment 1 Rocker 2 BCIP NBT 3 tray Animate the above figure with audio narration. 4 BCIP is the alkaline phosphatase substrate, which is dephosphorylated by the enzyme and then dimerizes. This dimer reduces nitroblue tetrazolim (NBT) to form an insoluble dark blue diformazan precipitate. Alkaline phosphatase is commonly conjugated to secondary antibodies. 5