The Greenhouse Effect and Global Warming
210 likes | 1.33k Vues
0. The Greenhouse Effect and Global Warming. Please take your assigned transmitter and swipe your student ID for attendance tracking. 0. The Temperature Structure of Earth’s Atmosphere. Exosphere: Heated by UV and X-rays from space. Thermosphere: Heated by X-rays from space.

The Greenhouse Effect and Global Warming
E N D
Presentation Transcript
0 The Greenhouse Effect and Global Warming Please take your assigned transmitter and swipe your student ID for attendance tracking.
0 The Temperature Structure of Earth’s Atmosphere Exosphere: Heated by UV and X-rays from space Thermosphere: Heated by X-rays from space Stratosphere: Heated by UV radiation from space Top of Ozone Layer Ozone Layer Troposphere: Heated by greenhouse effect Atmosphere gets colder at larger distance from heat sources.
:10 0 of 5 Which type of radiation from the sun will not be absorbed in the Earth’s atmosphere and can thus heat the Earth’s surface? • Infrared radiation. • Visible light. • Ultraviolet radiation. • X-rays. • Gamma-rays.
:10 0 of 5 In which form is the Earth giving off heat energy? • Infrared radiation. • Visible light. • Ultraviolet radiation. • Convection. • Mechanical energy.
:10 0 of 5 What happens to the infrared radiation re-emitted by the Earth’s surface in the troposphere? • Nothing. It will escape into space. • It is reflected back onto the Earth’s surface. • It gets scattered several times before it escapes into space. • It is absorbed. • It is transformed back into visible light.
The Greenhouse Effect 0 Earth’s surface is heated by the sun’s (visible) light. Heat energy is re-radiated from Earth’s surface as infrared radiation. CO2, but also other gases in the atmosphere, absorb infrared light → Heat is trapped in the atmosphere.
:10 0 of 5 Which of the following is another greenhouse gas (in addition to CO2)? • Oxygen (O2). • Nitrogen (N2). • Water vapor (H2O). • Sulfuric acid (H2SO4). • Hydrogen (H2).
0 Greenhouse Gases Gases consisting of compounds of two or more different types of atoms: • CO2 (carbon dioxide) • H2O (water vapor) • CH4 (methane) • NH3 (Ammonia) Most common gases in Earth’s atmosphere (N2, O2) do not contribute to the greenhouse effect.
:10 0 of 5 Does the greenhouse effect occur naturally? • Yes. • No.
The Greenhouse Effect 0 The Greenhouse Effect occurs naturally and is essential to maintain a comfortable temperature on Earth, but human activity, in particular CO2 emissions from cars and industrial plants, is drastically increasing the concentration of greenhouse gases.
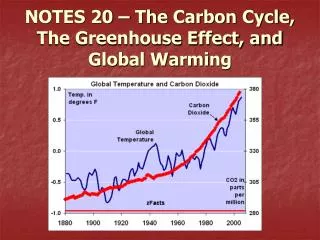
0 Global Warming • Human activity (CO2 emissions + deforestation) is drastically increasing the concentration of greenhouse gases. • As a consequence, beyond any reasonable doubt, the average temperature on Earth is increasing. • This is called Global Warming
:10 0 of 5 Which was the hottest year on history since 1880? • 1900 • 1952 • 1975 • 1999 • 2005
0 Consequences of Global Warming • Melting of glaciers and polar ice caps(→ rising sea water levels; rivers and fresh-water reservoirs will dry out!) Glacier National Park: Melting Glaciers 1911 Melting ice cap at Antarctica (South Pole): Temperature increase by 4.5 0F since 1945 2000
0 Consequences of Global Warming • Global climate changes: Extended droughts; El Niño Dry lake near Orlando, Florida Alaska: Permafrost subsiding: Warming by 3.5 0F since 1960
0 Consequences of Global Warming • Impacts on plant and animal life Dwindling penguin populations in Antarctica Olympic Mountains: Tree populations invading mountain meadow
0 Consequences of Global Warming • Impacts on plant and animal life Dying coral reefs
:10 0 of 5 In which atmospheric layer is ultraviolet radiation from space primarily absorbed? • The Troposphere • The Stratosphere • The Thermosphere • The Exosphere • The Magnetosphere
:10 0 of 5 Which molecule in the atmosphere is mainly responsible for the absorption of ultraviolet radiation from space? • Normal Oxygen (O2) • Ozone (O3) • Nitrogen (N2) • Hydrogen (H2) • Carbon dioxide (CO2)
:10 0 of 5 Which compound (used in some industrial processes like refrigeration and air conditioning) is destroying Ozone in the atmosphere? • Lead • CFCs • Water • Sulfuric Acid • Sulfur Dioxide
:10 0 of 5 The destruction of the ozone layer is currently most obvious above … • Central North America. • The North Pole. • The South Pole. • Central Asia. • Africa.
0 The Destruction of the Ozone Layer Ozone (= O3) absorbs UV radiation (which has damaging effects on human and animal tissue), causing the heating of the stratosphere. Chlorofluorocarbons (CFCs) (used, e.g., in industrial processes, refrigeration and air conditioning) destroy the Ozone layer. Destruction of the ozone layer as a consequence of human activity is proven (e.g., growing ozone hole above the Antarctic); Must be stopped and reversed by reducing CFC use, especially in developed countries!