Atom Review
430 likes | 549 Vues
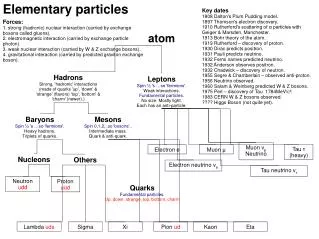
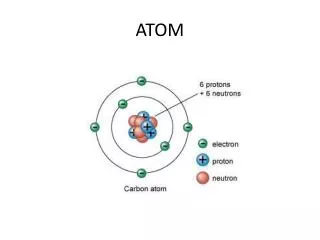
Atoms are the fundamental units of matter, each retaining the unique properties of an element. They consist of protons, which have a positive charge, and neutrons, which are neutral, located in the nucleus, while electrons, with a negative charge, orbit the nucleus in shells. The electron shell configuration is often summarized by the magic numbers: 2, 8, 18, and 32. The identity of an element is largely determined by the number of protons. This overview explores the characteristics and structure of atoms, including the role of subatomic particles.

Atom Review
E N D
Presentation Transcript
The smallest part of an element that retains the properties of the element is the ?
What is the magic # for the electron shells? • Bonus points: +5: what are the other shell numbers?
2,8,8 for the 1st 18. • 2, 8, 18, 32,50, 72
The charge of the nucleus of an atom is due to the presence of
121 • 201 • -80 121
When determining an elements identity, what is the MOST important subatomic part to examine?
If you have many models of an atom, how do you determine which one has the lowest mass?
Find the mass of this atom. • 23 P • 15 N
38 • 23+15= 38
No charge • About same mass as a proton
What is the charge of an atom’s nucleus if it has • 17 protons, 20 neutrons, and 17 electrons
Developing the model of an atom as a build up of successive orbital shells of electrons. • The model we use today.
12 • Mg • 24 • 12 P, 12 N, 12 E • 2 e, 8 e, 2 e in last shell