Chapter 2: Measurement and Problem Solving
16 days. Chapter 2: Measurement and Problem Solving. Day One. First day of school Lab safety. Syllabus HIGHLIGHTS!!!. Expectations Be Prepared NO FOOD OR DRINK IN THIS CLASSROOM EVER!!! How do I get a good grade? Hall Passes Absences. MISCELLANEOUS.

Chapter 2: Measurement and Problem Solving
E N D
Presentation Transcript
16 days Chapter 2: Measurement and Problem Solving
Day One First day of school Lab safety
Syllabus HIGHLIGHTS!!! • Expectations • Be Prepared • NO FOOD OR DRINK IN THIS CLASSROOM EVER!!! • How do I get a good grade? • Hall Passes • Absences
MISCELLANEOUS • BOOKS: Put name in front cover and please cover the book! • Three ring binder • Calculator • Email • Lab Safety Contract
Lab Notebook • Labs • Spiral bound notebook • Lab procedure: found online and in the room • Must be written in your lab notebook before you begin lab as well as any data tables you may need • Completed with a partner but must be a collaborative effort • Each partner is responsible to have the data before they leave the room
Location of Safety Equipment • Goggles • Aprons • Sand • Eyewash station • Safety Shower • MSDS Sheets • Fire Blanket • Fire extinguisher • Emergency Stop Button • Fire Alarm • Fume hood • Broken Glass Container Assignment: Draw a diagram of the room and label the safety equipment listed above. Explain what each of the above is used for and when they are used.
Day Two WebsitesMeasurements & Scientific Notation
Two websites you will be using all year!!! • www.nordoniahonorschemistry.wikispaces.com • Class website • Lab manual • Calendar • Lecture Notes • Answer Keys to Homework • www.masteringchemistry.com • Additional Reviews and assignments
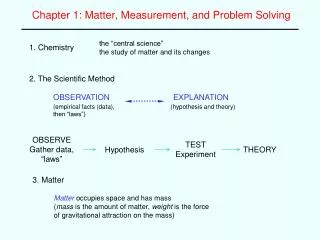
What Is a Measurement? • Quantitative observation • Comparison to an agreed upon standard • Every measurement has a number and a unit
A Measurement • The unit tells you to what standard you are comparing your object. • The number tells you: • What multiple of the standard the object measures. • The uncertainty in the measurement.
Scientists have measured the average global temperature rise over the past century to be0.6 °C • °C tells you that the temperature is being compared to the Celsius temperature scale. • 0.6 tells you that: • The average temperature rise is 0.6 times the standard unit of 1 degree Celsius. • The confidence in the measurement is such that we are certain the measurement is between 0.5 and 0.7 °C.
Scientific Notation • We should all be familiar with this but I just want to remind you of a few things!!! • S.N. is an easy way to express VERY large or VERY small numbers • Chemistry most of the time deals with extremely small number considering we talk about atoms!
3 parts to S.N. -9 1.2 10 X
How to Express a number in Scientific Notation • Move the decimal point to obtain a number between 1 and 10. • Write the result from Step 1 multiplied by 10 raised to the number of places you moved the decimal ** The exponent is POSITIVE if you moved the decimal to the LEFT ** The exponent if NEGATIVEif you moved the decimal to the RIGHT
Practice—Write the Following in Scientific Notation 123.4 145000 25.25 1.45 8.0012 0.00234 0.0123 0.000 008706
Practice—Write the Following in Standard Form 2.1 x 103 9.66 x 10-4 6.04 x 10-2 4.02 x 100 3.3 x 101 1.2 x 100
Day Three Metric and Factor Label
Metric System • Group of units used to make any kind of measurements • Best known for its simpleness • Why is it so simple? • Used all around the world with scientists
The Standard Units • Scientists generally report results in an agreed upon International System. • The SI System • Aka Système International
Common Prefixes in the SI System Tro's "Introductory Chemistry", Chapter 2
Let’s Try A Few • 125 cm = ____________ mm • 0.2568 L = _________ mL • 2.56 kg = ________ g
Factor LabelDimensional Analysis • EASY way to convert from one unit to another unit!! • WE will be doing this ALL YEAR long! So you better pay attention now! • UNITS ARE EXTREMELY IMPORTANT
Units • Always write every number with its associated unit. • Always include units in your calculations. • You can do the same kind of operations on units as you can with numbers. • cm × cm = cm2 • cm + cm = cm • cm ÷ cm = 1 • Using units as a guide to problem solving is called dimensional analysis.
Problem Solving and Dimensional Analysis • Many problems in chemistry involve using relationships to convert one unit of measurement to another. • Conversion factors are relationships between two units. • May be exact or measured. • Conversion factors generated from equivalence statements. • e.g., 1 inch = 2.54 cm can give or
Problem Solving and Dimensional Analysis, Continued • Arrange conversion factors so the starting unit cancels. • Arrange conversion factor so the starting unit is on the bottom of the conversion factor. • May string conversion factors. • So we do not need to know every relationship, as long as we can find something else the starting and desired units are related to :
Systematic Approach • Write down the given amount and unit. • Write down what you want to find and unit. 3. Write down needed conversion factors or equations.
Lets take a look at one! • I drive 20 miles to school every day. I want to know how many kilometers I drive.
Lets Try a Few • 125 lb = ________ kg • 2.5 gallons = ________ L • 3.1 km = _________ miles
Day Four Significant Figures and Factor label
Exact Numbers vs. Measurements • Sometimes you can determine an exact value for a quality of an object. • Often by counting. • Pennies in a pile. • Sometimes by definition • 1 ounce is exactly 1/16th of 1 pound. • Whenever you use an instrument to compare a quality of an object to a standard, there is uncertainty in the comparison.
45.872 Estimated Certain Reporting Measurements • Measurements are written to indicate the uncertainty in the measurement. • The system of writing measurements we use is called significant figures. • When writing measurements, all the digits written are known with certainty except the last one, which is an estimate. 45.872
Skillbuilder 2.3—Reporting the Right Number of Digits • A thermometer used to measure the temperature of a backyard hot tub is shown to the right. What is the temperature reading to the correct number of digits?
Reporting Numbers with Sig. Figs • When we report numbers in this class we need to make sure that we do not report them more specific than they really are. WHAT??? Volume of a box Length = 7.12 cm Width = 2.15 cm Height = 2.15 cm
How do I know if a number is significant? • All NONZERO digits are significant • Zeros between two nonzero numbers are significant • Trailing zeros are significant • Leading zeros are NOT significant, they are only place holders 5. Zeros at the end of a number but before a decimal are NOT significant
Lets try a few • 0.0035 • 1.080 • 2371 • 2.97 x 105 • 100000 • 0.500 • 58.31
Factor Label (Multi-step) • I went to the Yukon last summer and the speed limit sign said 105 km/hr. What was the speed limit in mi/hr?
If I drive a total of 50 miles a day and my car gets 49 miles/gallon. How much does it cost me a week to drive to work? Assuming price for gas is $2.50/gallon.
A circle has an area of 2,659 cm2. what is its area in square meters?
Day Five Significant Figures and Factor Label
Significant Figures in CalculationsADDITION AND SUBTRACTION • The difference and sum carries the same number of decimal places as the quantity carrying the FEWEST decimal places. • 3.449 cm – 0.76 cm = • 2.5 cm + 2 cm =
Significant Figures in Calculations(MULTIPLICATION AND DIVISION) • The results carries the same number of significant figures as the factor with the FEWEST figures. 5.892 cm X 6.10 cm =
Lets take a look back at our Volume of a Box!! Volume of a box Length = 7.12 cm Width = 2.15 cm Height = 2.15 cm
Lets Try a Few! • 1.01mm x 0.12 cm x 53.51 cm = • 56.55 cm x 0.920 cm = • 345.0 ml / 120 ml =
Day Six Significant Figures Lab
Day Seven Mastering Chemistry Website