Electron Energy Levels and Configurations
330 likes | 558 Vues
This unit covers the fundamentals of electron energy levels, their configurations, and orbital diagrams vital for understanding atomic structure. Key topics include Thomson's discovery of electrons, Bohr's energy level model, and the quantum mechanical model describing electron "clouds." We explore the principal energy levels (n=1 to n=7), the maximum number of electrons each level can hold, and the arrangement of sublevels and orbitals. Through practice with electron configurations and Lewis dot diagrams, students will grasp how valence and core electrons contribute to bonding.

Electron Energy Levels and Configurations
E N D
Presentation Transcript
Electron Energy Levels and Configurations Unit 6 Part 2 Unit Test: Wednesday January 15
Review of Atomic Theory • Thomsondiscovered the electron • Bohr determined that the electrons travel around the nucleus in energy levels; electrons with lower energy are closer to the nucleus • Quantum Mechanical Model says electrons orbit the nucleus in “clouds”; there is a high probability of finding them somewhere along these paths
Electrons: • are located at specific distances from the nucleus • Are found to have different amounts of energy
Principal Energy Levels • Only 7 principal energy levels are necessary • The main energy levels are numbered • start with n = 1 nearest the nucleus and go up to n = 7 farthest from the nucleus
The maximum number of electrons that can occupy a specific energy level can be calculated • Formula is 2n2 n = energy level • Calculate electrons for the 3rd energy level: • 2(3)2 = 18 electrons in the 3rd level
Principal Energy Levels • The first energy level holds a maximum of two electrons. • The second energy level is larger because it is farther away from the nucleus. • It holds a maximum of eight electrons. • The third energy level is larger still and holds a maximum of 18 electrons.
1 2 3 4 5 6 7 Energy Levels (add to notes) • Electrons are restricted to an energy level. • Electrons occupy the lowest energy level possible because electrons are lazy! • On the periodic table, each period (horizontal row) is an energy level
Energy Sublevels • Electrons carve out space around the nucleus • These spaces or patterns are called sublevels • Sublevels contain the orbitals where electrons are located • The 4 sublevels are identified by the letters s, p, d and f
Orbitals (add to notes) • Each sublevel contains a different # of orbitals • Orbitals are where electrons are found 90% of the time • Only two electrons can occupy the same orbital • This is because they spin in opposite directions
Rules for Determining Sublevels • No more than two electrons can occupy one orbital Again, why is this? • An electron will occupy the lowest sublevel possible… because …..????
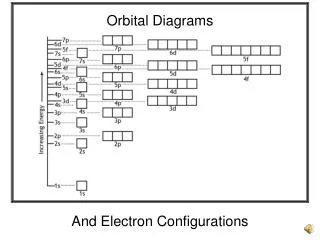
Locating Electrons • There are two ways to show where the electrons are found in the atom • Orbital filling diagrams • Electron configurations • Hund’s Rule -“Cupcake Rule” – everyone gets one before anyone gets two.
# electrons Helium: 1s2 Sublevel EnergyLevel Electron Configuration • Shorthand method for describing the arrangement of electrons • Composed of the principal energy level followed by the energy sublevel and includes a superscript with the # of electrons in the sublevel
Electron Configuration • Electron Configuration is ordered the way you read a book: from left to right and top to bottom • Note that d orbital is 1 energy level behind and the f orbital is 2 energy levels behind the s & p orbitals The order: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2 5f14 6d10 7p6 etc.
Practice • Hydrogen: • Oxygen: • Argon: • Copper: 1s1 1s1 2s2 2p4 1s2 2s2 2p6 3s2 3p6 1s2 2s2 2p6 3s2 3p6 4s2 3d9
Orbital Filling Diagrams • Each box represents an orbital which can hold a max of 2 e- • Aufbau principal – each electron occupies the lowest energy orbital available; German for “build up” • Electrons are notated with an arrow • Up arrow goes first, then down arrow • Arrows represent the opposing spin of electrons
Orbital Filling Diagrams • Hund’s Rule – when filling a sublevel, each orbital will contain 1 electron before a single orbital will contain 2 electrons • The three p orbitals fill in the order shown: • The number of arrows must match the number of electrons contained in the atom
Electron Configuration • Electron Configuration: 1s22s22p6 • Orbital Filling Diagram: • Orbital image:
Noble Gas Configuration • Shorthand electron configuration • Give the symbol of the noble gas in the previous energy level in brackets • Give the configuration for the remaining energy level • Example: Sulfur = 1s22s22p63s23p4 [Ne]3s23p4
Valence Electrons • Valence electrons: found in the outermost energy level • These electrons are used for bonding • Example: Nitrogen = 1s2 2s2 2p3 • Add up the number of e- (superscripts) • in the highest energy level • Nitrogen has 2 + 3 = 5 valence electrons • Core electrons: found in the inner energy levels. Nitrogen = 1s2 2s2 2p3
The number of valence electrons is equal to the number in the A group
Lewis-Dot Diagrams • Lewis Dot Diagrams are a way to represent the valence electrons in an atom. • Element’s symbol represents the nucleus and inner-level electrons • Dots represent the valence electrons • Dots are placed one at a time on the four sides of the symbol, then paired until all valence electrons are used… • Maximum of 8 e- will be around the symbol
Ions • Ions are atoms that have gained or lost electrons; indicated by a superscript that shows the ion’s charge Cl 1s22s22p63s23p5 Cl-1 1s22s22p63s23p6 chlorine added an electron
Chlorine adds a valence electron and becomes a negative ion. • Sodium loses a valence electron and becomes a pawsitive ion.
The ground state of an electron, the energy level it normally occupies, is the state of lowest energy for that electron. When an electron temporarily occupies an energy state greater than its ground state, it is in an excited state. An electron can become excited if it is given extra energy