Unit One: Classes of Elements, Periodic Table, and Stable Elements
E N D
Presentation Transcript
Notes One Unit One • Two Classes of Elements • Periodic Table Info? • What Are Stable Elements? • Stabilizing Sodium • Stabilizing Oxygen • Sodium Loses electrons to Oxygen • Oxidation Numbers • Key Elements and Examples
Two Classes of Elements • What are the Two Main Classes of Elements? • Metals and Nonmetals (Stair-Step)
What information doe the Periodic Table give us? Atomic Number # Protons = # of Electrons Electron structure Mass Number = # P + # N
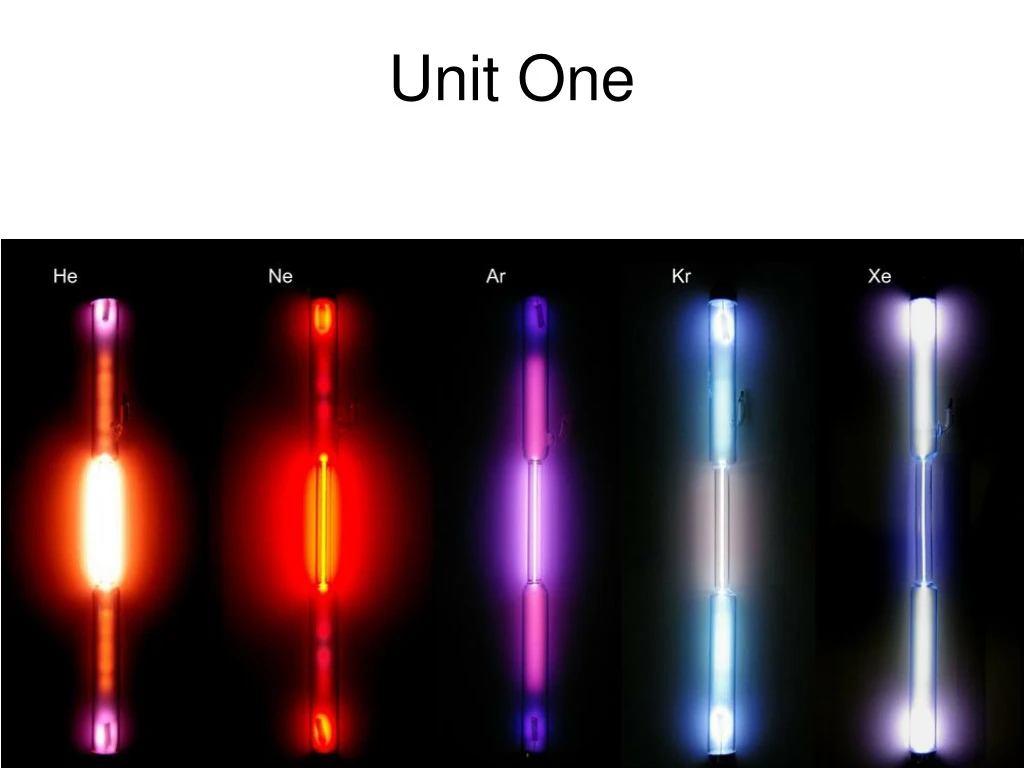
What Makes Elements Stable? Which orbitals? • Eight Electrons…… • Highest S and P…… • Noble Gases (electronically stable) • He, Ne, Ar, Kr, Xe, and Rn • Lose or Gain to get 8e-1 (to become stable) • Do metalsLose or Gain electrons? • Do nonmetalsLose or Gain electrons? Which Family? (Lose) (Gain)
Stabilizing Sodium • How many electrons does sodium have? • 11Na • What is Sodium’s Short-Hand Element? • 10Ne • How many Valence Electrons for Sodium? • 1e-1 • What is the shorthand electron configuration? • 11Na • Sodium loses/gain how many electrons? • 1e-1 • Na Na+1 + e-1 Oxidation or reduction? • (protons) + (electrons)=charge 1 [10Ne] 3s (+11) (-10) +1
Stabilizing Oxygen • How many electrons does oxygen have? • 8O • What is oxygen’s Short-Hand Element? • 2He • How many Valence Electrons for oxygen? • 6e-1 • What is the shorthand electron configuration? • 8O • Oxygen loses/gains how many electrons? • 2e-1 • O + 2e-1 O-2 Oxidation or reduction? • (protons) + (electrons)=charge 2p [2He] 2s 2 4 (+8) (-10) -2
Sodium Loses electrons to Oxygen • Na Na+1 + e-1 (Stable Like Neon) Ox or Red? • O + 2e-1 O-2 (Stable Like Neon) Ox or Red? • How many sodium atoms are needed to satisfy oxygen’s electron hunger? • 2e-1 means • How many oxygen atoms are needed to satisfy sodium’s electron loss? • 2e-1 means • Na2O High Electronegativity two Na Low Electronegativity One O
Oxidation Numbers • All elements Lose or Gain e-1. • Some have multiple loss or gain possibilities. S-2 Fe+2 Fe+3 S+4 S+6
Key Elements • (99%) H+1 H-1 • (99%)O-2 O-1 • (Always) Li+1, Na+1, K+1, Rb+1, Cs+1, Fr+1 • (Always) Be+2, Mg+2, Ca+2, Ba+2, Sr+2, Ra+2 • (Always) Al+3 • (with only a metal) F-1, Cl-1, Br-1, I-1 • (NO3-1) ion is always +5 • (SO4-2) ion is always +6
Example One • Find the oxidation numbers. • Al2S3 • Algebra is useful! • Al+3key element • 2(+3) + 3(S)=0 • S= -2 2(Al) + 0 3(S)=
Example Two • Find the oxidation numbers. • Ca(NO3)2 • Algebra is useful ! • Ca+2 and O-2key elements • (+2)+2(N)+6(-2)=0 • (+2)+2(N)+(-12)=0 • 2(N)+(-10)=0 • 2(N)=10 • N= +5 (Ca)+ 6(O)= 2(N)+ 0
Finding Oxidation #’s for Compounds +1 -2 +1 +4 -2 H2O H2CO3 +1 +5 -2 -3 +1 +4 -2 H3PO4 (NH4)2CO3 -2 +2 +5 +1 +5 -2 Ca3(AsO4)2 HNO3 +1 -2 +3 +6 +6 -2 H2SO4 Fe2(SO4)3 +1 +6 -2 +2 -2 +7 Hg2SO4 Ba(ClO4)2 +3 +4 -2 +6 +1 -2 Na2Cr2O7 Al2(CO3)3
Now it is time for class work !!! • A101: paper practice (Work Together) • CA101 : computer assignment • Both will be due at beginning of class next time • Ready Set Break !!!
Notes Two Unit One • Naming Inorganic Salts • Example One • Example One Thinking • Example Two • Computer Assignment One
Naming Inorganic Salts Positive Negative • TWO parts to the name • 1) Cation • 2) Anion • Cation Examples • Ca+2 • Al+3 • Fe+2 • Na+1 • Anion Examples • Cl-1 • NO3-1 • SO4-2 • N-3
Example One • Name the formula Fe2(CrO4)3 • Step #1 Find The + Ion(s). Iron(II) Fe+2 Iron(III) Fe+3
Example One • Step #2 Find The - Ion(s) Chromate CrO4-2
Example One Iron(II) Fe+3 Iron(III) CrO4-2 Fe2(CrO4)3 Fe+2 Chromate Chromate Chromate Iron(II) Iron(III) Fe+2 Fe+3 CrO4-2 CrO4-2 X (+2) + Y = 0 X (+3) + Y = 0 (-2) (-2) X=1 Y=1 X=2 Y=3 1 (+2) + 1 = 0 2 (+3) + 3 = 0 (-2) (-2) Fe Fe2(CrO4)3 CrO4
Example Two Al+3 Aluminum CO3-2 Al2(CO3)3 Carbonate Carbonate Aluminum Al+3 CO3-2 X (+3) + Y = 0 (-2) X=2 Y=3 2 (+3) + 3 = 0 (-2) Al2(CO3)3
Computer Assignment One/Two • NAMING IONIC COMPOUNDS LEVELs ONE AND TWO
(Cation+?)X(Anion-?)Y X (+?) + Y = 0 (-?) Writing a Formula From a Name If X orY is 2 or greater... Lowest Whole Number Ratio and the ion is polyatomic. Ba+2 Cr2O7-2 Hg2+2 Pb+4 ( )_( )_ Lithium Nitrate Li+1 1 NO3-1 1 LiNO3 ( )_( )_ Hydrogen Phosphate H3PO4 H+1 3 PO4-3 1 ( )_( )_ Ammonium carbonate NH4+1 CO3-2 1 2 (NH4)2CO3 ( )_( )_ Calcium Arsenate Ca3(AsO4)2 2 Ca+2 3 AsO4-3 ( )_( )_ Iron(III) periodate Fe+3 1 IO4-1 3 Fe(IO4)3 ( )_( )_ Mercury(I) Sulfate Hg2SO4 Hg2+2 2 SO4-2 2 ( )_( )_ Barium Perchlorate Ba+2 ClO4-1 2 1 Ba(ClO4)2 ( )_( )_ Sodium Dichromate Na2Cr2O7 1 Na+1 2 Cr2O7-2 ( )_( )_ Lead(IV) Sulfate Pb(SO4)2 4 Pb+4 2 SO4-2
Notes Three Unit One • Standard Amounts • One Gopher • One Mole • Formula mass • Percent Composition • Empirical Formula
Standard Amounts • How many dollars is… • A) 120 pennies? • 1.2 dollars • B) 2 quarters? • 0.5 dollars • C) 15 nickels? • 0.75 dollars • How many dozens is… • D) 48 eggs? • 4 dozen • E)18 apple fritters • 1.5 dozen
One Gopher • One Gopher equals 12 items • What is the mass of one gopher of… • A) white beads? • 2.81g/G • B) blue beads? • 0.50g/G • C) orange Beads? • 1.67g/G
Seven Rows Eight Rows
One Gopher(12 items) • In Six groups • (1) How many gophers of beads are in…(2) How many beads are in…A) ___gB) ___gC) ___gD) ___gE) ___gF) ___g 2.00 48 beads 4.00G 3.49 15 beads 1.24G 2.51 18 beads 1.50G 27 beads 2.25G 3.75 42 beads 1.75 3.50G 5.82 2.07G 25 beads
One Mole • One mole is 6.022x10+23 items. • Each element on the period table has a mass per mole. N 14.0g 6.022x10+23atoms 16.0g 6.022x10+23atoms O 12.0g 6.022x10+23atoms C • How many moles are in each? • How many atoms are in each? N 7.0g =0.50m x6.022x10+23atoms/m ÷14.0g/m =3.01x10+23atoms O =0.25m ÷16.0g/m x6.022x10+23atoms/m 4.0g =1.51x10+23atoms C =1.50m 18.0g ÷12.0g/m x6.022x10+23atoms/m =9.03x10+23atoms
Calculations Bases on Chemical Formulas Formula mass (Molecular Mass or Gram-Formula Mass) Empirical Formula Percent Composition
Rounding Atomic Mass 12.0 C 12.011 55.8 Fe 55.847 16.0 O 15.9994 209.0 Bi 208.980837 39.1 K 39.0983 197.0 Au 196.96654 190.2 Os 190.23 24.3 Mg 24.3050 23.0 Na 22.98968
Formula Mass Example One Calculate the formula mass for 1 mole of C6H12O6. # E Mass 12.011 C 12.0 = 72.0 6 x 1.0079 H 12 x 1.0 = 12.0 15.9994 O 6 x 16.0 = 96.0 180.0g/mol How many molecules of C6H12O6 is 180.0g/mol? 6.022x10+23 molecules
Empirical Formula Example One What is the empirical (simplest) formula containing 36.8% N, 63.2% O? 1) Calculate moles of each element. 3) Write the formula Q Mass E Mass N 2.63 mol N 14.0067 36.8 g ÷ 14.0 = 15.9994 O 63.2 g ÷ 16.0 = 3.95 mol O 2) Calculate the lowest ratio. Moles E Lowest Ratio N 1.00 2.63 mol N ÷ 2.63 mol = O 3.95 mol O ÷ 2.63 mol = 1.50 X by 2 to get whole numbers N2O3
Percent Composition Example One 1)Calculate the formula mass for 1 mole of H2O # E Mass Calculate the percentage composition of H2O. H 2.0 2 x 1.0 = 1.0079 O 1 x 16.0 = 16.0 15.9994 18.0g/mol 2) Divide each contribution by the total mass. ( x 100) = 11% H 0.11 2.0 ÷ 18.0 = Answer ( x 100) = 88.9% O 16.0 ÷ 18.0 = 0.889 3) Add the percentages to check work. 11% 88.9% 100.%
Empirical Formula Example Two What is the empirical (simplest) formula containing 69.58% Ba, 6.090% C, 24.32% O? 1) Calculate moles of each element. 3) Write the formula Q Mass E Mass Ba 0.50666 mol Ba 69.58 g ÷ 137.33 = C 6.090 g ÷ 12.01 = 0.50708 mol C O 24.32 g ÷ 16.00 = 1.520 mol O 2) Calculate the lowest ratio. E Moles Lowest Ratio Ba 0.50666 mol ÷ 1.000 0.50666 mol = C 0.50708 mol ÷ 0.50666 mol = 1.001 O 1.520 mol ÷ 0.50666 mol = 3.00 X by 1 to get whole numbers BaCO3
Percent Composition Example Two 1)Calculate the formula mass for 1 mole of Fe(ClO4)3. # E Mass Calculate the percentage composition of Fe(ClO4)3. 55.847 Fe 55.8 1 x 55.8 = 35.453 Cl 106.5 3 x 35.5 = 15.9994 12 x O 16.0 = 192.0 354.3g/mol 2) Divide the each contribution by the total mass. ( x 100) = 15.8% Fe 0.1575 55.8 ÷ 354.3 = Cl 0.3006 106.5 ÷ 354.3 = ( x 100) = 30.06% Answer O 192.0 ÷ 354.3 = ( x 100) = 54.19% 0.5419 3) Add the percentages to check work. 15.8 % 30.1 % 54.2 % 100.1%
Formula Mass Example Three Calculate the formula mass for 1 mole of Al2O3 # E Mass Al 54.0 2 x 27.0 = 26.98154 O 3 x 16.0 = 48.0 15.9994 102.0g/mol How many molecules of Al2O3 is102.0g/mol? 6.022x10+23 molecules
Formula Mass Example Two Calculate the formula mass for 1 mole of CaCO3. # E Mass 40.078 Ca 40.1 = 40.1 1 x 12.011 C 1 x 12.0 = 12.0 15.9994 O 3 x 16.0 = 48.0 100.1g/mol How many molecules of CaCO3 is100.1g/mol? 6.022x10+23 molecules