Boosted PI therapy: theoretical basis
This document discusses the theoretical foundation of Boosted Protease Inhibitor (PI) therapy and its influence on drug concentration, treatment-limiting toxicity, and therapeutic windows in HIV management. It includes critical data from various studies, highlighting the efficacy thresholds, viral rebound effects, and the relationship between drug adherence and virologic outcomes. By exploring interventions like directly observed therapy (DOT), this work aims to draw insights into optimizing treatment regimens and improving patient responses to antiretroviral therapy for better health outcomes.

Boosted PI therapy: theoretical basis
E N D
Presentation Transcript
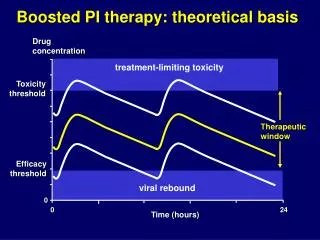
Boosted PI therapy: theoretical basis Drug concentration treatment-limiting toxicity Toxicity threshold Therapeuticwindow Efficacy threshold viral rebound 0 0 24 Time (hours)
MaxCmin1 - HIV RNA <400 c/ml ITT - Exposed 100 saquinavir/r (n=148) 80 % patients indinavir/r (n=158) 60 40 20 0 Baseline 24 weeks Dragstead UB et al. 8th ECCATH, Athens, 2001. Abst O10
Male 30 yr, HIV+ 1994, ARC 1995 CD4=300 AZT 3TC IDV D4T ddI NVP AZT 3TC EFV ddI ABC SQV LPV/r Switch EFV to DLV HIV RNA log10 c/ml CD4 cells/mm3 Dual nucs HyperS stop ABC switch to Comb Comb ddI EFV LPV/r SQV APV 400 1000000 100000 300 10000 PCP 200 1000 100 100 Sept 99 June 00 Oct 00 Feb 01 June 01 Sept 01
Clinical progression on PI based Tx by CD4 and HIV RNA response (n=2236) All patients 100 CD4 RNA 95 CD4 RNA CD4 RNA 90 % without new AIDS event 85 CD4 RNA 80 75 6 9 12 15 18 21 24 27 30 PI Treatment time (months) Grabar et al. Ann Int Med2000;133:401-419
OT ITT Virologic efficacy of HAARTOn-treatment versus intent-to-treat analyses Loss in efficacy because of poor tolerability and adherence 100 28 80 35 43 52 60 % patients <400 copies/ml at 48 wks 40 20 0 Abacavir CNAAB30052 Indinavir CNAAB30052 Amprenavir PROAB30013 Efavirenz Study 0061 1. Staszewski et al.N Engl J Med 1999;341:1865-1873 2.Staszewski et al.JAMA 2001;285:1155-1163 3. Goodgame et al.Antiviral Ther 2000;5:215-225
Directly observed therapy (DOT) and RNA decline Prisoners in 4 clinical trials by DOT or self-administration (SAT) 100 DOT SAT 80 P<0.01 60 HIV RNA <50 copies/ml 40 20 0 wk 4 wk 8 wk 16 wk 24 wk 32 wk 40 wk 48 Fischl M et al. 7th CROI 2000. San Francisco: Poster 71
MaxCmin1 – Pill count and viral load response at week 24 (ITT, nc=f) 100 80 Total12 PI PillsPer Day 60 % patients <400 copies/ml Total6 PI PillsPer Day 40 20 0 Indinavir/r (n=159) Saquinavir/r (n=158) Adapted from Dragstead UB et al. 8th ECCATH, Athens, 2001. Abstract O10 and Presentation
“Relative to my other HIV/AIDS drugs,injections have not limited or altered my ability to...” Moderate activities 96 Prepare meals 95 Vigorous activities 91 Family life 89 Social relationships 87 Perform work 78 Be intimate w/partner 76 Perform daily activities 62 Personal appearance 62 Privacy of health 62 Travel 54 0 20 40 60 80 100 % of patients who agree (somewhat or strongly) Cohen C et al.1st IAS Conference on HIV Pathogenesis and Treatment, Buenos Aires, 2001, Poster 708
Inside – investigational drugs 2001 RT inhibitors Nucleosides DAPD Emtricitabine (FTC) d4C ACH-126,443 BCH-10618 Nucleotides Tenofovir (PMPA) NNRTIs Emivirine TMC 120 and 125 Calanolide A DPC 083 DPC 961 DABO compounds
Inside – investigational drugs 2001 Protease inhibitors Tipranavir Atazanavir (BMS 232632) Mozenavir (DMP-450) DPC 681/684 Tibotec compound (TMC 114) Fos-amprenavir (GW433908) • Other Inhibitors • Integrase inhibitors: L-drugs, S-1360 • RNase H inhibitors • Zinc finger inhibitors • Others
Outside – Investigational drugs 2001 Entry inhibitors Attachment PRO 542, Pro 367, FP-21399, sCD4 Co-receptor CXCR4: ALX40-YC, Met-SDF-1, KRH-120, T-22, AMD 3100 CCR5:SCH-C, SCH-D, PRO 140, TAK 779, RANTES derivatives Fusion T-20, T-1249, 5 helix, d-peptides
Atazanavir boosts saquinavir 10000 1600 SQV/100 RTV qd 1000 SQV concentration (ng/ml) 1600 SQV/400 TAZ qd 1200 SQV tid 100 1200 SQV/400 TAZ qd 10 0 5 10 15 20 25 Time (hours) TAZ exposure unaffected by SQV coadministration Saag et al. ICAAC 1999, Abs 330 O’Mara et al. 7th CROI 2000, Abs 504
T-20: efficacy in ARV-experienced patients at 48 weeks Percent of patients responding 56% copies/ml 60 <400 or >1 log10 50 D from baseline 40 33% 30 20 10 0 All patients who completed 48 weeks (n = 41) Intent-to-treat (n = 70) *Intent-to-treat (ITT) analysis: non-completer = failure Lalezari et al. 13th International AIDS Conference, Durban, 2000, Abs LbPp116
T1249 active against T-20 resistant variants 120 100 80 60 % of untreated control virus 40 T20 (day 28) 20 T1249 (day 28) T20 (day 0) 0 T1249 (day 0) 0.001 0.01 0.1 1 10 Concentration (g/ml) TRI003 Patient #13 Lambert D et al. Antiviral Therapy 1999. 4; (Suppl 1);8