Copper Minerals
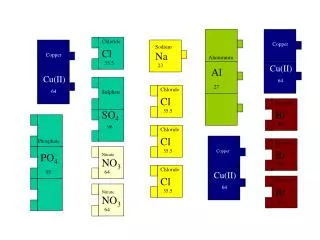
Copper Minerals. a % composition exercise. Simple Copper Sulfides. Chalcocite: copper (I) sulfide. Tenorite: copper (II) sulfide. 1. Write the formulas for each sulfide and determine the %Cu. Complex Copper Sulfides. Bornite Cu 5 FeS 4. Chalcopyrite CuFeS 2.

Copper Minerals
E N D
Presentation Transcript
Copper Minerals a % composition exercise
Simple Copper Sulfides Chalcocite: copper (I) sulfide Tenorite: copper (II) sulfide 1. Write the formulas for each sulfide and determine the %Cu.
Complex Copper Sulfides Bornite Cu5FeS4 Chalcopyrite CuFeS2 2. From inspection (by looking), which ore has the highest %Cu? Calculate the %Cu in each to see if you’re right!
Semiprecious Copper Gems Malachite CuCO3.Cu(OH)2 Azurite 2CuCO3.Cu(OH)2 3a. Determine the %Cu in each gem. 3b. Which contains more copper: 10.0 g of azurite or 9.00 g of malachite?
Cuprite: copper (I) oxide While most copper compounds are blues or greens, there is one red one. single crystal more usual form (microcrystalline) 4a. Find the %Cu in cuprite. 4b. How many Cu atoms are there in a 5.25 g crystal of cuprite? 4c. If a sample contained 0.30 moles of Cu atoms, how many moles of oxygen atoms would it have?
Answer Key copper (I) sulfide = Cu2S; copper (II) sulfide = CuS % Cu in Cu2S = {(2*63.5 g)/[(2*63.5 g)+32.1 g]}*100 = 79.8% % Cu in CuS = [(63.5 g)/(63.5 +32.1 g)]*100 = 66.4% 2. With 5 Cu, bornite looks to have a higher %Cu than chalcopyrite with one Cu. bornite = 63.3% Cu chalcopyrite = 34.6% Cu 3a. azurite (3Cu, 8O, 2H, 2C)= 55.3% Cu malachite (2Cu, 1C, 5O, 2H) = 57.5% Cu 3b. 10.0 g azurite * 0.553 = 5.53 g Cu 9.00 g malachite *0.575 = 5.18 g Cu, so AZURITE HAS MORE Cu 4a. copper (I) oxide = Cu2O; cuprite = 88.8% Cu 4b. 5.25 g sample* 0.888 = 4.66 g Cu 4.66 g Cu * (1 mole/63.5 g) * (6.02 x 1023 atoms/1 mole) = 4.42 x 1022 atoms 4c. Cu2O has one oxygen for every 2 copper atoms, so a sample with 0.30 mol. Cu would have (1/2)(0.30 mol) = 0.15 moles oxygen