Periodic Table Day 2
150 likes | 242 Vues
Learn about atoms, matter, elements, compounds, and mixtures. Discover types of mixtures, properties of matter, and common elements using chemical formulas.

Periodic Table Day 2
E N D
Presentation Transcript
Periodic Table Day 2 Mrs. King – 7th Grade Science
HUSTLE – • What is an atom? • What is matter? • What is an element? • What is a compound? • What is a mixture? • What are 2 types of mixtures? Explain. • What are the 2 properties of matter? • Give examples of both.
Goals/Standards • Standards:. • 7-5.2: Classify matter as element, compound, or mixture on the basis of its composition. • 7-5.5: Translate chemical symbols and the chemical formulas of common substances to show the component parts of the substances • Goal: • We will review mixtures and determine the difference between elements and compounds.
Elements/Compounds – Page 17 • ELEMENTS • Pure substances that cannot be changed into simpler substances • Composed of only 1 kind of atom • COMPOUNDS • Pure substances that are composed of 2 or more elements (Chemically combined) • Compounds can only be changed into simpler substances called elements by chemical changes • One way that 2 or more atoms can combine is to form a molecule
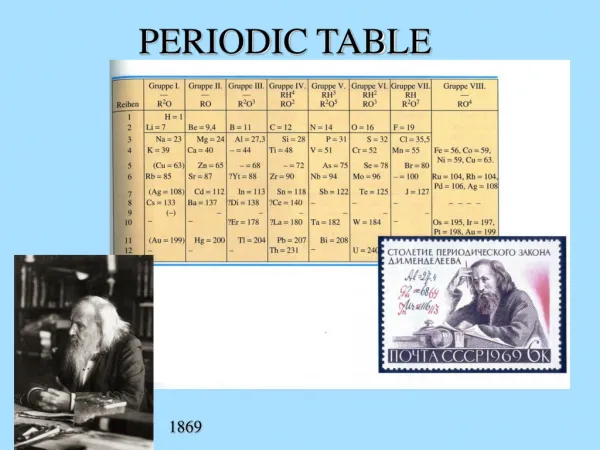
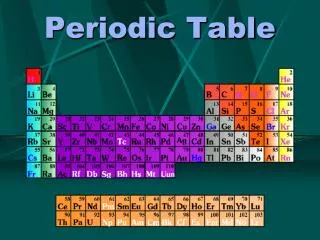
The elements on the periodic table are arranged numerically by atomic numbers. • Families, also called groups,are vertical columns of elements on the periodic table; they are usually numbered 1-18. Elements in the same family have similar properties.
A Few More Pieces of Info • Chemical symbols show the atoms of the element that it is composed of. • Symbols are written with 1, 2 or 3 letters • The first letter is ALWAYS capitalized. • Each element has a different symbol.
On the periodic table there is a zigzag line on the right side of the table. There are two sections of elements on the periodic table, metals and nonmetals.
Metals • A major classification of elements generally located on the left side of the zigzag line on the periodic table. • Examples of metals are: Sodium (Na), Calcium (Ca), Iron (Fe), and Aluminum (Al). The majority of elements are metals.
Nonmetals • A major classification of elements generally located on the right side of the zigzag line on the periodic table. • Examples of nonmetals are: Chlorine (Cl), Oxygen (O), Sulfur (S), and Iodine (I).
Compounds Continued • Compounds are composed of more than 1 element. • The formulas have more than 1 type of symbol showing the different elements that compose the compound.
Chemical Formulas • Constructed from the symbols of the elements composing the substances. • The # of subscripts show how many of each kind of atom are in the compound. • The subscript is written to the lower right of the element symbol. • If no subscript is written, only 1 atom of that element is part of the compound.
What are some Elements that we use NaCl [salt] H2O [water] C6H12O6 [simple sugar], O2 [oxygen gas] CO2 [carbon dioxide N2 [nitrogen gas])
Whew…that was a LOT! • Let’s Go Back Over It! • Elements? • Compounds? • Chemical Formulas? • Help me out!
PASS Challenge • Sugar is a solid made of carbon, hydrogen, and oxygen atoms. Hydrogen and oxygen are gases at room temperature. Sugar is • A. an element • B. a compound • C. a homogeneous mixture • D. a heterogeneous mixture