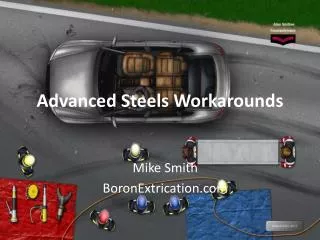
Quenched Steels
Quenched Steels. A quenched steel is composed of: Martensite – dark needles Retained austenite – light matrix regions in micrograph. A quench steel is subject to residual stresses caused by: Uneven contraction due to sudden hardening of the exterior

Quenched Steels
E N D
Presentation Transcript
Quenched Steels A quenched steel is composed of: • Martensite – dark needles • Retained austenite – light matrix regions in micrograph
A quench steel is subject to residual stresses caused by: • Uneven contraction due to sudden hardening of the exterior • Expansion of the interior, which later transforms to martensite • The transformation expansion of austenite to martensite is greater than the thermal contraction. Quenched Steels
Quenched Steels If the stress exceeds the yield point, then the plastic deformation causes warping. If the stress exceeds the fracture stress then quench cracks will occur at the surface martempering or marquenching – the austenite is cooled to just above Ms and held until the temperature equalizes in the steel. Subsequent quenching permits all the steel to transform to martensite at about the same time. This prevents quench cracks to form at the surface due to the surface transforming to martensite before the interior of the steel, which puts the surface in tension above the yield strength and the center in compression.
Properties of Quenched Steels Martensite and retained austenite are both metastable phases and break down to form more stable constituents when a steel is aged, ie., held for a long period at low temperature (few hundred of degrees to room temperature). As the phases formed during ageing involve changes in specific volume, quenched martensitic steels exhibit dimensional changes over a period of time, which renders them unsuitable for a large number of applications. In addition, although martensitic steels are harder than pearlite steels, they are also considerably more brittle.
Properties of Quenched Steels The combination of dimensional instability and brittleness results in increased quench cracking, which again makes quenched steels unsuitable for structural engineering applications. Adding alloying elements thatlower Ms (C > Mn > Mo > Cr > Si > Ni > Cu ) increase the incidence of warpage and quench cracking because of the increase in retained austenite. High carbon steels are also more susceptible to quench cracking because of the increased hardness of the martensite. Quenched steels are thus given a heat treatment, known as tempering (~200 oC), to eliminate the dimensional changes and induce toughness.
The martensitic reaction in carbon steels is NOT reversible at low temperatures. When a quenched steel is heated at a temperature below the eutectoid, it will be converted to a mixture of intermediate metastable phases (eg. e-carbide, bainite), or, if the temperature is high enough, it will transform to stable ferrite plus cementite. Tempering of Quenched Steels (pearlite) (bainite)
Hardness Changes During Tempering Hardness is increased at temperatures from RT to 100 oC due to clustering ofcarbon at defects and the “coherent” precipitation of e-carbide. The hardnessdrops above 200 oC due to a lowering of the carbon content in the martensite by the precipitation of rods of cementite, but in contrast to e–carbide, this precipitation is “not coherent”.
Hardness Changes During Tempering The hardness falls further at higher tempering temperatures but at a reduced rate due to two parallel processes: 1) Spheroidization and later particle growth of cementite 2) Recovery, and later recrystallization, of ferrite (formed from martensite) occurs.
Precipitation of e–Carbide during Tempering In the image below, freshly quenched martensite is not visible as they have tilted surface facets, which are under oblique incident illumination in the microscope. Fine dispersion of e–carbide formed by tempering below 100 oCare not resolved in the light microscope, but cause the martensite needles to become darkened. Tempered martensite at 500x mag
Formation and Growth of Cementite during Tempering Rod-like structures of cementite after 1 hr at 250 oC Spheroidized cementite formed after 1 hr at 500 oC Globular particle growth of cementite after 12 hrs at 675 oC
Effect of Carbon Content and Tempering Temperature on Hardness of Martensite The hardness values plotted against carbon content refer to the hardness of quenched martensite after tempering for 1 hr at various temperatures. These hardness changes cover a wider range of C composition than seen in the earlier slide.
Effect of Alloying Elements on Hardness of Tempered Martensite All elements other than C in solution slow down the softening of a steel during tempering. The alloying elements may enter either the ferrite or the carbide phase in a tempered steel structure, i.e., “partitioning”. Elements such as Al, Cu, Si, P, Ni, Zr, do not form stable carbide phases and are thus foundonly in the ferrite phase. Other elements such as Ti > V > Mo > W > Cr > Mn form stable carbides, based on cementite, with the formula (Fe M)3C. These precipitates are found in both the ferrite and carbide phases.
Tetragonal Fe3C Structure - carbon (Ti, V, Mo, W, Cr, Mn)* - Fe * - these elements also partition into the Fe3C phase.
Effect of Alloying Elements on Hardness of Tempered Martensite A high temperature hardening effect is observed at higher temperatures > 540 C, when carbide forming elements are present, due to the formation of much harder carbides of the type M6C23. This results in the secondary hardening as seen with Mo.
Quench and Tempered Treatments The mechanical properties of a high carbon steel that is quenched and tempered to produce a fine dispersion of cementite in a ferrite matrix are superior to the mechanical properties of a rapidly cooled steel with a fine pearlite structure. By selecting an appropriate tempering temperature, a wide range of properties can be produced.
Quench and Temper Treatments The effect of tempering temperature on the properties of a quenched 1050 steel is shown below.
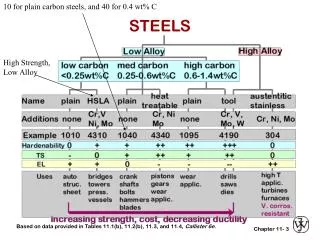
Classification of Carbon Steels (Not Stainless Steels) There are four main groups of carbon steels, which can be defined by their carbon concentration. • Low carbon steels (> 80% of the steel production) < 0.20 %C Used in normalized condition for structural purposes. 2. Medium Carbon Steels 0.25 – 0.55 %C Used in heat treated conditions for machine parts. 3. Eutectoids Steels 0.6 – 0.8 %C Used in normalized condition for hard wearing rails. 4. Tools Steels 0.9 – 2.2 %C Used in heat treated condition for tools and dies.
Classification of Carbon Steels (Not Stainless Steels) This is where we start talking about specific steel alloys. We will discuss these four broad categories over the next couple of lectures.
Plain-carbon steels are designated most commonly by the American Iron and Steel Institute (AISI) or the Society of Automotive Engineers (SAE) A four digit code is used: If the first two digits are 10, the steel is plain carbon steel. Other designations, ie., 20s, 30s, etc. refer to major alloying additions (see next slide). The last two digits indicate the nominal carbon content of the steel in hundredths of a percent. Ferrous Alloy Charts
AISI Designation for SteelsWe will be discussing the properties of many of these steels.
American Society for Testing Materials (ASTM) Specifications Steels for defined structural purposes (buildings, bridges, etc.) are usually made to ASTM specifications, which use a system of numbering that is unrelated to chemical composition, mechanical properties, or applications. The numbers simply refer to the chronological order in which the steels were developed and approved for inclusion in theASTM system.
Group 1 Steels: Low Carbon Structural Steels The bulk for the steels used for industrial processes are plain carbon steels that contain less than 0.20 %C, with microstructures of ~80% ferrite and ~20% pearlite. In order to minimize processing costs, these steels are not heat treated. It is not practical to heat treat very large sections used for bridges, ships and buildings. Their strength is thus controlled by: 1) The carbon content 2) The grain size (via the Petch-Hall relationship s = so + kd-1/2) As these steels are susceptible to the ductile-brittle transitions at room temperature, the Mn content is increased to improve toughness.
Controlled Hot Rolling Programs to Obtain Fine Grain Size Special rolling procedures are used to obtain a very fine grain size, ~ 5 mm, in low carbon steels in the absence of a heat treatment. The final hot rolling pass is performed around 828 oC, at which temperature recrystallization of austenite does not occur, so that the austenite grains are left in a deformed state at 727 oC. Further, since the austenite grains are flattened by the rolling deformation, the total austenite grain boundary area is increased, leading to an increased number of ferrite nucleation sites, which in turn decreases the ferrite grain size. The effect of hot rolling to improve strength by lowering the austenite grain size is illustrated in the next two slides for a 0.2 %C steel.
Ferrite Controlled Hot Rolling Programs to Obtain Fine Grain Size The additional increase in strength obtained by using alloying elements is shown by the 0.2 %C HSLA steel (High-Strength-Low-Alloy (< 10% alloying additions), which was given the same treatment.
Microalloyed Non-Heat Treatable Low Carbon Steels In a hot rolled plain carbon steel, austenite can recrystallize at temperatures as low as 760 oC, ie., at only 33 oC above the eutectoid temperature. To reduce the work involved, hot rolling is performed at temperatures > 760 oC so recrystallization of austenite and subsequent “grain growth is the norm in hot rolled plain carbon steels”. The time required to recrystallize austenite is greatly increased by the addition of a little as 0.05 % of carbide forming elements such as V, Ti and Nb.
Microalloyed Non-Heat Treatable Low Carbon Steels • The effect is shown for 0.1 %C steels rolled to 50 %RA at 954 oC with: • The addition of 2.0 %Mn, which is a non-carbide former • Additions of 0.03 %Nb + 0.00, 0.05, 0.20 %V, which are strong carbide formers. • The carbide formers form fine particles of carbonnitrides – eg., NbCN – which 1) pin the grain boundaries and 2) prevent recrystallization, which 3) prevents grain growth. Fe-0.1%C-2%Mn-0.03%Nb + …
Thus the alloying additions that are carbide formers can be used as another tool by engineers to control the strength and ductility of the steel. In many instances, the steels are already strong enough for the application. Ductility or fabricability is desired. Microalloyed Non-Heat Treatable Low Carbon Steels
Thin Steel Sheets for Automobile Bodies Automobile body stock is produced either from “rimmed steel” (not deoxidized) or from “killed steel” (deoxidized with Al) – expressions used by industry. The killing action also provides a method to control and reduce the grain size. It gives lower strength but with increased elongation or ductility. These general steels contain 0.05 – 0.15 %C and 0.2 – 0.6 %Mn with impurities level of: 0.001 % (max) Si, 0.05 %(max) S and 0.02 %(max) P.
Thin Steel Sheets for Automobile Bodies These steels have to be cold-formed into the desired shape and yet retain a good surface finish, which requires a combination of low yield and high tensile strength with a high degree of uniform elongation. The grain size is also critical (< ASTM 9 impairs cold forming and > ASTM 7 gives an orange peel surface). A sharp yield point means non-uniform elongation, which causes surface roughness. So the sheet is tempered rolled to about 1% reduction by “roller flexing”, which stretches each surface beyond the yield point, before cold forming.
Tin Plate The American automotive industry now extensively uses Sn-plated plain C sheet steel forautobodies to reduce corrosion. Steel for tin plate contains 0.12 %(max) C, 0.2 – 0.6 %Mn, .01 %(max) Si, 0.05%(max)S, 0.02 %(max) P and 0.2 %(max) Cu – essentially “The Tin Can” material. The steel is first hot rolled and then cold rolled to the finished gauge. After rolling it is process-annealed in coils and then tempered-rolled and then electrolytically plated with tin.
When a tin-can competition occurred with Al and plastic containers, the cost of tin plate was reduced by annealingafter rolling such that a further 35% cold roll gave the final gauge, which enabled thicknesses of 0.125 mm to be obtained, enabling it to have better mechanical properties than Al and plastics. Although this treatment increases the yield pointto within 7 MPa of the UTS and the final measured tensile elongationis less than 1%, the sheet is still sufficiently ductileto flange the ends of can bodies without fracture. The reason for this apparent contradiction is that the tensile elongation is measured over a gauge length of 2 inches (50 mm), but the stretching just before fracture is localized to a much smaller length of material, which makes it possible. If a gauge length of 0.01 inches (0.25 mm) was used, the measured elongation would be 40 %. (see next page) Tin Plate
Tin Plate Note the yield and UTS are almost the same.
Zinc Plated Low Carbon Steels(galvanized steel) • If Tin cracks, it’ll establish an electrolytic cell with the steel and it will corrode the steel faster than the steel will corrode by itself since Tin is a more noble metal than steel. • For some applications, a more effective steel coating is Zinc, which is less noble than steel but more difficult to coat because of its low vapor pressure. • At one time, galvanized steel pipes were preferred for a home’s water system, however, they were only good for 20-30 years and homes last much longer. • They have now been replaced by Copper, which lasts longer and is more flexible.
Zinc Plated Low Carbon Steels(galvanized steel) • In 1999, Dofasco started up its 72"-wide hot dipped galvanizing line joint venture with Arcelor of France. • This hot dip coating technology now produces 450,000 tons per year of corrosion-resistant galvanized steel primarily for automotive applications and with a particular focus on exposed auto body panels. • This product is a cost-effective replacement for electrogalvanized material for exposed automotive applications. • This is another reason why Dofasco is capturing the America automobile market for steel and American/Canadian made cars are improving over foreign made cars. What else did Dofasco develop that revolutionized the steel industry?
Steels for thick plates and pressure vessels contain: 0.18 – 0.35 %C, 0.06 – 1.25 %Mn and impurity levels of: 0.15 – 0.35 %Si, 0.04 %(max) S and 0.035 %(max) P The higher Mn level gives improved strength without lowering ductility and Cu is sometimes added to improve corrosion resistance. These steels are usually given no heat treatment after hot rolling, apart from a normalizing treatment for sections greater than 5 cm in thickness. Heavy Steel Plates for Bridges and Tanks Can you recall a normalizing heat treat?
A1 is the eutectoid transformation temperature. A3 is the ferrite proeutectoid temperature of formation. Acm is the cementite proeuctectoid temperature of formation. Summary of simple heat treatments for hypoeutectoid and hypereutectoid steels.
Heavy Steel Plates for Bridges and Tanks These steels have only moderate strength, but their particular combinations of strength, ductility, toughness and “weldability” are required for fabricating massive structures, eg., for ship building. A lower ductile-brittle transition temperature for low temperature applications is obtained by: 1) using higher Mn content killed steels, 2) performing extensive hot work, and by 3) normalizing.
The effect of carbon and heat treatment on the mechanical properties of plain-carbon steels.
Heavy Steel Plates for Buildings, Bridges and Pressure Vessels Hence, for improved low temperature toughness, steels such as A516 contains more Mnthan A515 and is specified to be in the normalized condition with a grain size < ASTM 5 (i.e., 16 grains per inch2 at 100x magnification). Steel A662 contains more Mn and less carbon and retains its toughness down to -60 oC. “70” refers to the strength of the steel in kips per square inch (ksp), an old strength designation still used in the USA.
Heavy Steel Plates for Buildings and Bridges White grains are ferrite and dark regions are pearlite colonies. A516 Grade 70 Heavy Plate Steel Ferrite and fine pearlite occur after normalizing from 900 oC. The low carbon and alloy content of this steel also restricts the microstructure to ferrite and pearlite in quenched sections that are > 5 cm thick.