Chapter 20 Organic Chemistry
Chemistry: A Molecular Approach , 1 st Ed. Nivaldo Tro. Chapter 20 Organic Chemistry. Roy Kennedy Massachusetts Bay Community College Wellesley Hills, MA. 2008, Prentice Hall. Structure Determines Properties. Organic compounds all contain carbon

Chapter 20 Organic Chemistry
E N D
Presentation Transcript
Chemistry: A Molecular Approach, 1st Ed.Nivaldo Tro Chapter 20OrganicChemistry Roy Kennedy Massachusetts Bay Community College Wellesley Hills, MA 2008, Prentice Hall
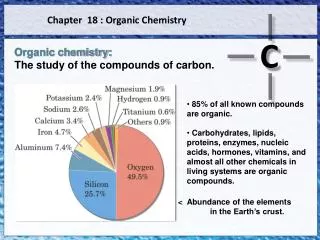
Structure Determines Properties • Organic compounds all contain carbon • CO, CO2 , carbonates and carbides are inorganic • other common elements are H, O, N, (P, S) • Carbon has versatile bonding patterns • chains, rings, multiple bonds • chain length nearly limitless • Carbon compounds generally covalent • molecular; gases, liquids, or low melting solids; varying solubilities; nonconductive in liquid • C - C bonds unreactive (very stable) Tro, Chemistry: A Molecular Approach
Allotropes of Carbon - Diamond Tro, Chemistry: A Molecular Approach
Allotropes of Carbon - Graphite Tro, Chemistry: A Molecular Approach
Carbon Bonding • mainly forms covalent bonds • C is most stable when it has 4 single covalent bonds, but does form double and triple bonds • C=C and C≡C are more reactive than C−C • C with 4 single bonds is tetrahedral, • 2 singles and 1 double is trigonal planar • 2 doubles or 1 triple and 1 single is linear Tro, Chemistry: A Molecular Approach
Hydrocarbons • hydrocarbons contain only C and H • aliphatic or aromatic • insoluble in water • no polar bonds to attract water molecules • aliphatic hydrocarbons • saturated or unsaturated aliphatics • saturated = alkanes, unsaturated = alkenes or alkynes • may be chains or rings • chains may be straight or branched • aromatic hydrocarbons Tro, Chemistry: A Molecular Approach
Saturated Hydrocarbons • a saturated hydrocarbon has all C-C single bonds • it is saturated with hydrogens • saturated aliphatic hydrocarbons are called alkanes • chain alkanes have the general formula CnH2n+2 Tro, Chemistry: A Molecular Approach
Unsaturated Hydrocarbons • unsaturated hydrocarbons have one of more C=C double bonds or CC triple bonds • unsaturated aliphatic hydrocarbons that contain C=C are called alkenes • the general formula of a monounsaturated chain alkene is CnH2n • remove 2 more H for each additional unsaturation • unsaturated aliphatic hydrocarbons that contain CC are called alkynes • the general formula of a monounsaturated chain alkyne is CnH2n-2 • remove 4 more H for each additional unsaturation Tro, Chemistry: A Molecular Approach
Unsaturated Hydrocarbons Tro, Chemistry: A Molecular Approach
Aromatic Hydrocarbons • contain benzene ring structure • even though they are often drawn with C=C, they do not behave like alkenes Tro, Chemistry: A Molecular Approach
alkanes alkenes alkynes Tro, Chemistry: A Molecular Approach
Formulas • molecular formulas just tell you what kinds of atoms are in the molecule, but they don’t tell you how they are attached • structural formulas show you the attachment pattern in the molecule • models not only show you the attachment pattern, but give you an idea about the shape of the molecule
Condensed Structural Formulas • attached atoms listed in order • central atom with attached atoms • follow normal bonding patterns • use to determine position of multiple bonds • () used to indicate more than 1 identical group attached to same previous central atom • unless () group listed first in which case attached to next central atom Tro, Chemistry: A Molecular Approach
Line-Angle Formulas • each angle, and beginning and end represent a C atom • H omitted on C • included on functional groups • multiple bonds indicated • double line is double bond, triple line is triple bond Tro, Chemistry: A Molecular Approach
Formulas Tro, Chemistry: A Molecular Approach
Formulas Tro, Chemistry: A Molecular Approach
Isomerism • Isomers = different molecules with the same molecular formula • Structural Isomers = different pattern of atom attachment • Constitutional Isomers • Stereoisomers = same atom attachments, different spatial orientation Tro, Chemistry: A Molecular Approach
Structural Isomers of C4H10 Butane, BP = 0°C Isobutane, BP = -12°C Tro, Chemistry: A Molecular Approach
Rotation about a bond is not isomerism Tro, Chemistry: A Molecular Approach
Possible Structural Isomers Tro, Chemistry: A Molecular Approach
Ex 20.1 – Write the structural formula and carbon skeleton formula for C6H14 Tro, Chemistry: A Molecular Approach
Ex 20.1 – Write the structural formula and carbon skeleton formula for C6H14
Ex 20.1 – Write the structural formula and carbon skeleton formula for C6H14
Stereoisomers • stereoisomers are different molecules whose atoms are connected in the same order, but have a different spatial direction • optical isomers are molecules that are nonsuperimposable mirror images of each other • geometric isomers are stereoisomers that are not optical isomers Tro, Chemistry: A Molecular Approach
Nonsuperimposable Mirror Images mirror image cannot be rotated so all its atoms align with the same atoms of the original molecule Tro, Chemistry: A Molecular Approach
Chirality • any molecule with a nonsuperimposable mirror image is said to be chiral • any carbon with 4 different substituents will be a chiral center • a pair of nonsuperimposable mirror images are called a pair of enantiomers Tro, Chemistry: A Molecular Approach
Optical Isomers of 3-methylhexane Tro, Chemistry: A Molecular Approach
Plane Polarized Light • light that has been filtered so that only those waves traveling in a single plane are allowed through Tro, Chemistry: A Molecular Approach
Optical Activity • a pair of enantiomers have all the same physical properties except one – the direction they rotate the plane of plane polarized light • each will rotate the plane the same amount, but in opposite directions • dextrorotatory = rotate to the right • levorotatory = rotate to the left • an equimolar mixture of the pair is called a racemic mixture • rotations cancel, so no net rotation Tro, Chemistry: A Molecular Approach
Chemical Behavior of Enantiomers • a pair of enantiomers will have the same chemical reactivity in a non-chiral environment • but in a chiral environment they may exhibit different behaviors • enzyme selection of one enantiomer of a pair Tro, Chemistry: A Molecular Approach
Alkanes • aka paraffins • aliphatic • general formula CnH2n+2for chains • very unreactive • come in chains or/and rings • CH3 groups at ends of chains, CH2 groups in the middle • chains may be straight or branched • saturated • branched or unbranched Tro, Chemistry: A Molecular Approach
Naming • each name consists of 3 parts • prefix • indicates position, number, and type of branches • indicates position, number, and type of each functional group • parent • indicates the length of the longest carbon chain or ring • suffix • indicates the type of hydrocarbon • ane, ene, yne • certain functional groups Tro, Chemistry: A Molecular Approach
Naming Alkanes • Find the longest continuous carbon chain • Number the chain from end closest to a branch • if first branches equal distance use next in • Name branches as alkyl groups • locate each branch by preceding its name with the carbon number on the chain • List branches alphabetically • do not count n-, sec-, t-, count iso • Use prefix if more than one of same group present • di, tri, tetra, penta, hexa • do not count in alphabetizing
H C H - , M E T H Y L H C 3 H H H C H C H - , E T H Y L H C C 3 2 H H H H H C H C H C H - , P R O P Y L H C C C 3 2 2 H H H H C H 3 ( C H ) C H - , I S O P R O P Y L H C C 3 2 H H Alkyl Groups Tro, Chemistry: A Molecular Approach
H H H H C H C H C H C H - , n - B U T Y L H C C C C 3 2 2 2 H H H H H H C H 3 C H C H ( C H ) C H - , s e c - B U T Y L H C C C 3 2 3 H H H H C H H 3 ( C H ) C H C H - , I S O B U T Y L H C C C 3 2 2 H H H C H 3 B U T Y L H C C ( C H ) C - , tert- - 3 3 3 C H 3 More Alkyl Groups Tro, Chemistry: A Molecular Approach
H H H H H H C C C C C H H CH3 H H H H CH3 H H H H H C C C C C C H H CH3 CH H H H CH3 CH3 Examples of Naming Alkanes 2-methylpentane 3-isopropyl-2,2-dimethylhexane Tro, Chemistry: A Molecular Approach
Example – Name the alkane • find the longest continuous C chain and use it to determine the base name since the longest chain has 5 C the base name is pentane Tro, Chemistry: A Molecular Approach
Example – Name the alkane • identify the substituent branches there are 2 substituents both are 1 C chains, called methyl Tro, Chemistry: A Molecular Approach
Example – Name the alkane • number the chain from the end closest to a substituent branch • if first substituents equidistant from end, go to next substituent in then assign numbers to each substituent based on the number of the main chain C it’s attached to 1 2 3 4 5 both substituents are equidistant from the end 2 4 Tro, Chemistry: A Molecular Approach
Example – Name the alkane • write the name in the following order • substituent number of first alphabetical substituent followed by dash • substituent name of first alphabetical substituent followed by dash • if it’s the last substituent listed, no dash • use prefixes to indicate multiple identical substituents • repeat for other substituents alphabetically • name of main chain dimethyl pentane 2,4 – 2 4 Tro, Chemistry: A Molecular Approach
Practice – Name the Following Tro, Chemistry: A Molecular Approach
Practice – Name the Following 3-ethyl-2-methylpentane Tro, Chemistry: A Molecular Approach
1 2 3 4 5 6 Drawing Structural Formulas 4-ethyl-2-methylhexane • draw and number the base chain carbon skeleton • add the carbon skeletons of each substituent on the appropriate main chain C • add in required H’s Tro, Chemistry: A Molecular Approach
Practice – Draw the structural formula of 4-isopropyl-2-methylheptane Tro, Chemistry: A Molecular Approach
Practice – Draw the structural formula of 4-isopropyl-2-methylheptane Tro, Chemistry: A Molecular Approach
Alkenes • also known as olefins • aliphatic, unsaturated • C=C double bonds • formula for one double bond = CnH2n • subtract 2 H from alkane for each double bond • trigonal shape around C • flat • much more reactive than alkanes • polyunsaturated = many double bonds Tro, Chemistry: A Molecular Approach
Alkenes ethene = ethylene propene Tro, Chemistry: A Molecular Approach