Atom
Information about atom

Atom
E N D
Presentation Transcript
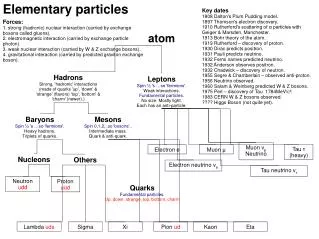
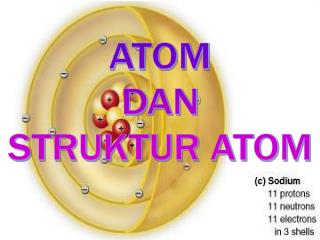
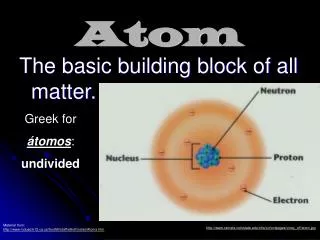
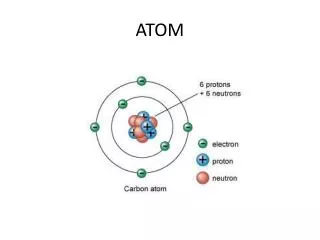
ATOM All matter consists of extremely small particles called atom . The subatomic particle of atom are • Electron • Proton • Neutron
DISCOVERY OF ELECTRON DISCHARGE TUBE EXPERIMENT • When electrons move in a straight line, such types of rays are called cathode ray and are produced when current is passed through a gas at 0.01 atmosphere to 0.0001 atmosphere pressure in a discharge tube . • These cathode rays are deflected toward positive plate when electric field is applied and on applying magnetic field these rays are deflect toward north pole.
DISCOVERY OF PROTON DISCHARGE TUBE EXPERIMENT • Anode rays are defined as positively charged rays produced in a discharge tube experiment when current is passed through the gas at low pressure . • These rays are deflected toward the negative plate when electric field is applied and on applying magnetic field these are deflected toward south pole of the magnet.
DISCOVERY OF NEUTRONS In 1932, James Chadwick discovered that most nuclei also contain another neutral particle called neutron, which is slightly more massive than proton but has no charge. His experiment involved bombarding Beryllium atoms with alpha particles, which produced a strong beam of particles that was not deflected by electrical field .
The first simple model of atom was proposed by J.J Thomsan. According to this model an atom is consists of a uniform sphere of positive charge in which electrons are embedded so that electrostatic force of attraction are maximum . This model looks like a watermelon in which seeds are embedded. Therefore, Thomson model of atom also known as watermelon or pudding model.
RUTHERFORD SCATTERING EXPERIMENTS RUTHERFORD ALPHA PARTICLE SCATTERING EXPERIMENT AND DEFLECTION OF ALPHA PARTICLES
Rutherford and his co-workers performed a series of experiment , they bombard alpha-particle on the thin gold foil and note following observation:- • Most of the alpha particle are passed through the gold foil undeflected. • A small fraction of alpha particle got deflected through small angles. • Very few (about one in 20,000) did not pass through the foil at all but suffer large deflection or even come back suffering a deflection of 180 0 .
BOHR’s MODEL OF ATOM • According to Bohr’s model of atom , an atom consits of postively charged nucleus in the centre around which electrons are revolving in a fixed circular path called orbit. • The energy of electron in the orbit does not change with time. • Only those orbit are permitted in which angular momentum is a whole number multiple of h/2Π. • The energy is emitted or absorbed only when the electrons jump from one energy level to another i.e. electron jump from ground state to excited state.