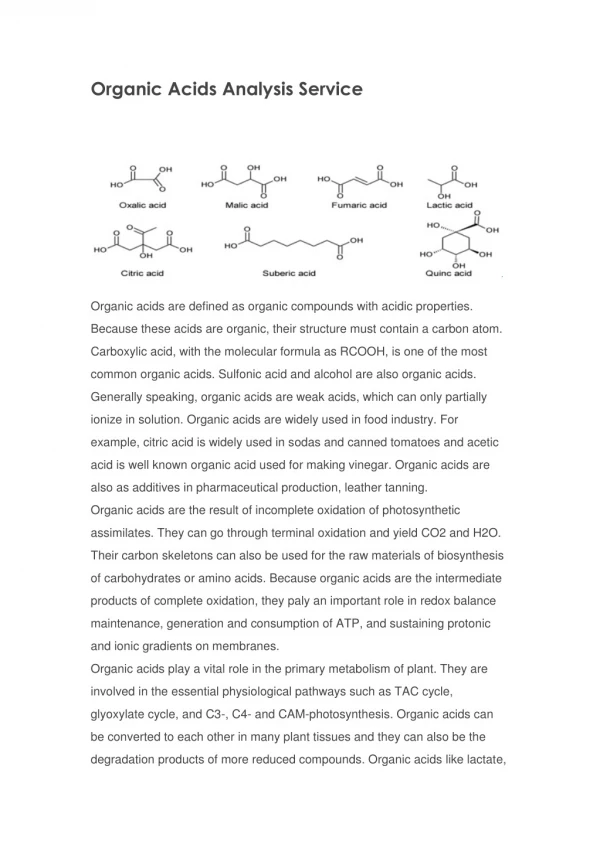
Organic Analysis
Organic Analysis. Chapter 5. OK – Some really basic chemistry. Matter – anything that has mass and occupies space. Element - The purest form of any matter. There are about 92 naturally occurring elements.


Organic Analysis
E N D
Presentation Transcript
Organic Analysis Chapter 5
OK – Some really basic chemistry • Matter – anything that has mass and occupies space. • Element - The purest form of any matter. • There are about 92 naturally occurring elements. • Atom - The smallest particle of an element that can exist without losing the properties of that element. (Meaning – if you breakdown an atom of nitrogen, you no longer have nitrogen). • Periodic Table – A chart that arranges elements in groups and series.
Combining Atoms • Molecule – a chemical combination of 2 or more atoms of the same element – like O2, N2, H2, etc. • Compound – contain atoms of 2 or more different elements – like caffeine (C8H10N4O2) , cocaine(C17H21NO4 ), etc. • There are 16 million known compounds in the universe (More to be discovered later!)
Back to Matter • Matter is made up of atoms and can exist in 4 different forms: • Solid – has definite shape and volume - is rigid. Molecules have fixed distances from each other. • Liquid – has volume, but takes on the shape of its container – it is not rigid, but fluid. Molecules are close, but moving and switching places with each other. • Gas – does not have a definite shape nor volume. Molecules are far apart and do not make much contact with each other. • Plasma – a type of gas, where some electrons have “come loose” from the atoms in the gas – so the gas atoms have become ions. That is why plasma is also known as “ionized gas”.
A huge, handle-shaped cloud of relatively cool dense plasma suspended in the sun's hot, thin corona. Natural Plasma Ball lightning
How can you determine what chemicals are in an unknown sample? • Perform a basic test on it – there are specific tests for known compounds – for example, a white powder can be tested to see if it is cocaine, or any other drug. • If a test is inconclusive, the unknown compound can be analyzed by “pulling it apart”. Individual chemical groups or molecules can be separated out of the compound.
Sedimentation and DECANTATION • A very quick method for separating a mixture of a liquid and a heavier solid. • If we want to separate a mixture of water and sand, we should allow the sand to settle on the bottom of the container. Then we pour out the water. • Advantage of this method: Fast, simple • Disadvantage of this method: Crude. It cannot be used to separate a mixture of a liquid and a light sold, such as chalk in water. The particles of chalk are suspended in the water. They are so light that they do not sink down to the bottom for a long time. This will also not work on solutions – where the solvent particle may never settle down.
Centrifugation (Forced sedimentation) and decantation • This involves using centrifugal force to cause small undissolved particles to settle down, thus separating components out. • Advantage – fast, easy • Disadvantage – cannot separate small, dissolved molecules – for example salt solution – remains a salt solution.
Filtration • This is a method which is effective for separating suspensions, for example mud in water. All one needs is filter paper or a sieve with different pore/hole sizes in it. • Advantages – fast, simple • Disadvantages – cannot separate small, dissolved particles or parts of compounds
Evaporation • Useful for separating dissolved compounds. For example, if salt water is heated, water molecules evaporate, leaving salt crystals behind. • Advantages – easy • Disadvantage – only solute can be isolated – the solvent is lost to evaporation
Distillation • Evaporation + Condensation = Distillation • Once the solvent evaporates, it isn’t lost – it is collected and cooled. • Advantage – can keep both – solute and solvent • Disadvantage – Cannot separate multiple solutes and solvents
Solution to Problem: Chromatography! • A procedure used to separate mixtures • How does it work? • You need 2 phases or media: a moving phase and a stationary phase. • You dissolve the mixture to be separated in the moving phase • You run the moving phase with dissolved materials over the solid phase • The dissolved chemicals that have a greater affinity to the stationary phase will be slowed down and are the last to leave • The dissolved chemicals with a lower affinity to the solid phase move over it at a greater rate and leave sooner
A Chromatography analogy • An analogy which is sometimes useful is to suppose a mixture of bees and wasps passing over a flower bed. The bees would be more attracted to the flowers than the wasps, and would become separated from them. If one were to observe at a point past the flower bed, the wasps would pass first, followed by the bees. In this analogy, the bees and wasps represent the analytes to be separated, the flowers represent the stationary phase, and the mobile phase could be thought of as the air.
Analytes (Bees and Wasps) Air (Moving Phase) Stationary Phase (Flowers) Wasps don’t Bees like flowers So wasps will leave first
Types of Chromatography • Gas chromatography • the moving phase is a carrier gas such as Helium or Nitrogen that moves through a column • The stationary phase is a thin film of liquid within the same column • As the analytes travel through the column, the molecules with greater affinity to the carrier gas will leave the column faster and those with greater affinity to the liquid film will leave later – thus the mixture gets separated into individual components • As each component emerges from the column, it passes through a detector. The Detector uses a flame to ionize the compound and to generate an electric signal • This signal is recorded and printed out onto a strip-chart recorder as a function of time – this is called a Chromatogram
A Chromatogram Relative abundance of analyte Retention Time (Min)
How to read a Chromatogram • The amount of analyte is determined by how high the peak is (Chromatography is extremely sensitive, it can detect very small concentrations – as low as 1 ng) • The type of analyte is determined by the time it emerged from the column – how many minutes.
Types of Chromatography • Liquid chromatography or High Performance Liquid Chromatography (HPLC) • The moving phase is a liquid • The stationary phase are solid particles
Types of Chromatography • Thin-Layer Chromatography • The Stationary phase is a solid, such as paper, or a plastic strip coated with a film of solid particles • The Moving phase is a liquid such as water, acetone, alcohol, etc. • The components with the greatest affinity to the moving liquid will travel faster If the separated components are not visible to the naked eye, UV light can be used to see if they fluoresce.
Mass Spectrometry • Is usually coupled with a Gas Chromatograph, because the GC is not always identify the chemical • In Mass Spectrometry, no 2 substances can produce the same pattern • The GC first, like before, separates the components • They then flow into the Mass Spec’s vacuum chamber. • Here, the MS converts the incoming molecules into positive ions by making them lose electrons – this makes them unstable • Some molecules even disintegrate • These fragments then enter an electric field which separates them according to their mass. • Since every atom or ion has a different and specific mass, it can produce a very specific line on the graph that is produced.
Gas Chromatography Time (min) Mass Spectrometry m/z (mass-to-charge ratio)
Electrophoresis • Similar to TLC – it separates materials according to their migration rates across a stationary phase. • But instead of using a liquid or gas as the moving phase, it uses an electric current. • In forensic science, it is most often used to separate proteins from dried blood and DNA fragments during DNA fingerprinting. • DNA fragments get separated according to their size (all DNA is negatively charged) • Proteins get separated according to: • Size • Charge
What is Electromagnetic Radiation? • Electromagnetic radiation can be described in terms of a stream of photons, which are massless particles (they actually have EXTREMELY LOW MASS) each traveling in a wave-like pattern and moving at the speed of light. • So, all electromagnetic radiation travels at the speed of light (c) which is 299,792,458 meters per second (1,079,252,848.8 km/h). • Each photon contains a certain amount (or bundle) of energy, and all electromagnetic radiation consists of these photons. • The only difference between the various types of electromagnetic radiation is the amount of energy found in the photons. • Radio waves have photons with low energies, microwaves have a little more energy than radio waves, infrared has still more, then visible, ultraviolet, X-rays, and ... the most energetic of all ... gamma-rays.
How can Electromagnetic Radiation affect us? • EMR comes from all kinds of different sources – the primary source being outer space. EMR also comes from the sun (all heavenly bodies), man-made objects such as radios, etc. • EM radiation carries energy and momentum, which may be transferred to any matter when it interacts with the matter. • For example, Ultraviolet radiation (from the sun) can cause damage to our DNA – resulting in skin cancer. • Microwave radiation agitates molecules of water, producing heat and eventually “cooking” the matter (hopefully food)
Are all the photons different? • All the photons are the same, they just contain different amounts of energy and therefore travel at different wavelengths • Radio waves, visible light, X-rays, and all the other parts of the electromagnetic spectrum are fundamentally the same thing. They are all electromagnetic radiation
The Visible Spectrum • Some photons have energy and wavelengths that allow us to see them • This is visible light – what is light? • Zillions of photons moving in a wave-like pattern with energy levels that make them visible to the human eye. • Some animals like fish and snakes, can see photons (wavelengths) that we cannot
Visible Light • So visible light appears white, but it is a collection of photons traveling at different wavelengths and frequencies. • You can split white light into its different wavelengths using a glass prism.
Coherent and Incoherent Light • Laser light has several features that are significantly different from white light. • White spreads out as it travels, so less light hits a surface as the distance from the light source increases. Laser light travels as a parallel beam and spreads very little. • White light is a jumble of colored light waves and incoherent. Laser light is monochromatic and coherent. • Incoherent – all the crests and troughs are NOT parallel • Coherent – all crests and troughs are parallel
Spectrophotometry • Using photons of visible light to measure and analyze materials. • You can use it to determine how much light a particular solution absorbs • You can also use it to determine how much light a particular solution transmits (allows to pass through) • You can also determine the wavelength of light the sample absorbed or transmitted.
Who uses it? • Visible region 400-700nm spectrophotometry is used extensively in colorimetry science. Ink manufacturers, printing companies, textiles vendors, and many more, need the data provided through colorimetry. • And ofcourse, forensic scientists use it!