Exploring the Law of Conservation of Matter through Halloween Chemistry
Dive into the chemistry of sodium and chlorine reactions to understand the Law of Conservation. Analyze results to explore matter creation and destruction. Homework and video review included.

Exploring the Law of Conservation of Matter through Halloween Chemistry
E N D
Presentation Transcript
Bell Ringer 10-15-13 • Mrs. Thomasis making a special Halloween concoction. She mixes 34.5 grams of sodium and 11.8 grams of chlorine. When she added the two together there was a chemical reaction that yielded 38.3 grams of sodium chloride (salt). Were these results accurate? Explain your answer.
MYP Unit Question: How can such a small thing impact our environment in such a big way? • Essential Question: Can matter be created or destroyed? • Area of interaction: Environment • Learner profile words: Communicators, inquirers • Standard:S8P1. Students will examine the scientific view of the nature of matter. • g. Identify and demonstrate the Law of Conservation of Matter. • Learning Target: Today I am learning about the Law of Conservation of Matter, this is important because what you start with is what you end with (at least in a chemical reaction). • Homework: Complete analysis questions for Lab, start study for end of the unit test 10-29-13
Agenda • Opening: • Password • Work Session: • GPB Video • Closing: • Exit Ticket
Opening – Password Rules • No yelling out • You cannot say rhymes with
Clock is… The ****Practice Word
Compound is… The ****Practice Word
Reactivity is… The ****Practice Word
Physical Property is… The ****Practice Word
The Law of Conservation of Matter is… The ****Practice Word
Indicators of a CHEMICAL CHANGE is… The ****Practice Word
Work Session • Complete the notes as you review the video and place on IAN pg.
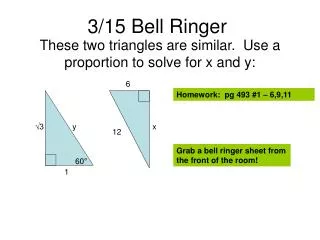
Closing- Exit ticket • Use the diagram below to answer questions