Exploring Carboxylic Acids: Properties and Uses
130 likes | 184 Vues
Learn about methanoic, ethanoic, propanoic, and butanoic acids, their physical properties, boiling points, occurrence in nature, and various industrial uses. Understand the solubility of short-chain acids and the significance of carboxylic dimers.

Exploring Carboxylic Acids: Properties and Uses
E N D
Presentation Transcript
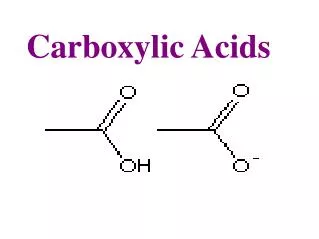
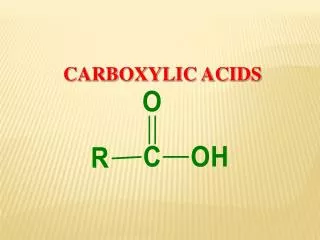
Carboxylic acids • Methanoic acid HCOOH • Ethanoic acid CH3COOH • Propanoic acid C2H5COOH • Butanoic acid C3H7COOH
Physical properties • Physical state: Methanoic acid and ethanoic acid are liquids, while propanoic acid and butanoic acid are solids • Short chain carboxylic acids are soluble in water due to the polar COOH group • Carboxylic acids are soluble in non-polar solvents such as cyclohexane
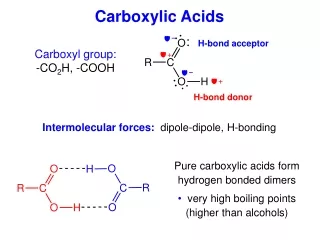
Boiling points of carboxylic acids • Boiling points higher than the corresponding alcohols • This is because carboxylic acids form dimers, where two carboxylic acid molecules are held together by two hydrogen bonds • This is possible due to polarity in both the C=O and O-H bonds in each carboxylic acid molecule
Ethanoic acid dimer δ- δ+ δ- δ+ δ+ δ- δ+ δ-
Occurrence and uses • Methanoic acid is found in the sting of ants and nettles • Ethanoic acid is the principal acid in vinegar • Ethanoic acid is used in the manufacture of cellulose acetate • Propanoic acid, benzoic acid and their salts (e.g. sodium benzoate) are used as food preservatives