Solids
This article provides a comprehensive overview of different types of solids, including ionic, covalent, molecular, and metallic. It also discusses the band theory, temperature dependence of resistivity, and the behavior of electrons in semiconductors.

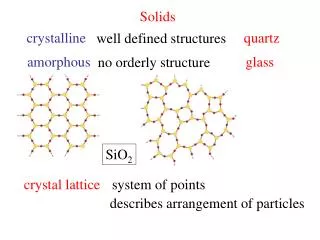
Solids
E N D
Presentation Transcript
Solids Eisberg & Resnick Ch 13 & 14 RNave: http://hyperphysics.phy-astr.gsu.edu/hbase/solcon.html#solcon Alison Baski: http://www.courses.vcu.edu/PHYS661/pdf/01SolidState041.ppt Carl Hepburn, “Britney Spear’s Guide to Semiconductor Physics”. http://britneyspears.ac/lasers.htm
http://hyperphysics.phy-astr.gsu.edu/hbase/solcon.html#solconhttp://hyperphysics.phy-astr.gsu.edu/hbase/solcon.html#solcon
OUTLINE • Review Ionic / Covalent Molecules • Types of Solids (ER 13.2) • Band Theory (ER 13.3-.4) • basic ideas • description based upon free electrons • descriptions based upon nearly-free electrons • ‘Free’ Electron Models (ER 13.5-.7) • Temperature Dependence of Resistivity (ER 14.1)
Ionic Bonds RNave, GSU at http://hyperphysics.phy-astr.gsu.edu/hbase/chemical/bond.html#c4
Ionic Bonding RNave, Georgia State Univ at hyperphysics.phy-astr.gsu.edu/hbase/molecule
Covalent Bonds RNave, GSU at http://hyperphysics.phy-astr.gsu.edu/hbase/chemical/bond.html#c4
Covalent Bonding SYM ASYM spatial spin ASYM SYM spatial spin space-symmetric tend to be closer
Covalent Bonding not really parallel, but spin-symmetric Stot = 1 Stot = 0 not really anti, but spin-asym space-symmetric tend to be closer
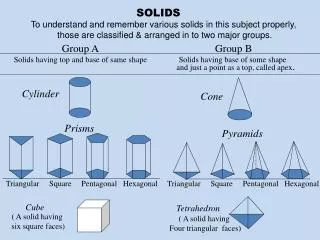
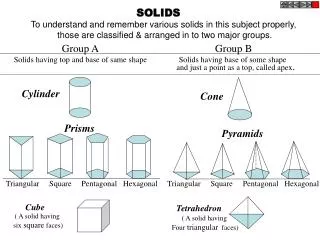
TYPES OF SOLIDS (ER 13.2)CRYSTALINE BINDING • molecular • ionic • covalent • metallic
most organics inert gases O2 N2 H2 Molecular Solids • orderly collection of molecules held together by v. d. Waals • gases solidify only at low Temps • easy to deform & compress • poor conductors
NaCl NaI KCl Ionic Solids • individ atoms act like closed-shell, spherical, therefore binding not so directional • arrangement so that minimize nrg for size of atoms • tight packed arrangement poor thermal conductors • no free electrons poor electrical conductors • strong forces hard & high melting points • lattice vibrations absorb in far IR • to excite electrons requires UV, so ~transparent visible
Ge Si diamond Covalent Solids • 3D collection of atoms bound by shared valence electrons • difficult to deform because bonds are directional • high melting points (b/c diff to deform) • no free electrons poor electrical conductors • most solids adsorb photons in visible opaque
Fe Ni Co Metallic Solids config dhalf full • (weaker version of covalent bonding) • constructed of atoms which have very weakly bound outer electron • large number of vacancies in orbital (not enough nrg available to form covalent bonds) • electrons roam around (electron gas ) • excellent conductors of heat & electricity • absorb IR, Vis, UV opaque
Six Closely Spaced Atomsas fn(R) the level of interest has the same nrg in each separated atom
Two atoms Six atoms Solid of N atoms ref: A.Baski, VCU 01SolidState041.ppt www.courses.vcu.edu/PHYS661/pdf/01SolidState041.ppt
Four Closely Spaced Atoms conduction band valence band
Solid composed of ~NA Na Atomsas fn(R) 1s22s22p63s1
Sodium Bands vs Separation Rohlf Fig 14-4 and Slater Phys Rev 45, 794 (1934)
Copper Bands vs Separation Rohlf Fig 14-6 and Kutter Phys Rev 48, 664 (1935)
Differences down a column in the Periodic Table: IV-A Elements same valence config Sandin
Band Spacingsin Insulators & Conductors electrons free to roam electrons confined to small region RNave: http://hyperphysics.phy-astr.gsu.edu/hbase/solcon.html#solcon
How to choose eFandBehavior of the Fermi function at band gaps
How does one choose/know eF If in unfilled band, eF is energy of highest energy electrons at T=0. If in filled band with gap to next band, eF is at the middle of gap.
FermionsT=0 RNave: http://hyperphysics.phy-astr.gsu.edu/hbase/solcon.html#solcon
Number of Electrons at an Energy e In QStat, we were doing Number of ways to have a particular energy distrib fn Number of electrons at energy e
# states probability of this nrg occurring # electrons at a given nrg
Semiconductors ~1/40 eV • Types • Intrinsic – by thermal excitation or high nrg photon • Photoconductive – excitation by VIS-red or IR • Extrinsic – by doping • n-type • p-type ~1 eV
Intrinsic Semiconductors Silicon Germanium RNave: http://hyperphysics.phy-astr.gsu.edu/hbase/solcon.html#solcon
Doped Semiconductors lattice p-type dopants n-type dopants
5A doping in a 4A lattice 5A in 4A lattice 3A in 4A lattice
5A in 4A lattice 3A in 4A lattice
‘Free-Electron’ Models • Free Electron Model (ER 13-5) • Nearly-Free Electron Model (ER13-6,-7) • Version 1 – SP221 • Version 2 – SP324 • Version 3 – SP425 • .
********************************************************* • Free-Electron Model • Spatial Wavefunctions • Energy of the Electrons • Fermi Energy • Density of States dN/dE E&R 13.5 • Number of States as fn NRG E&R 13.5 • Nearly-Free Electron Model (Periodic Lattice Effects) – v2 E&R 13.6 • Nearly-Free Electron Model (Periodic Lattice Effects) – v3 E&R 13.6
Free-Electron Model (ER13-5) classical description
Quantum Mechanical Viewpoint In a 3D slab of metal, e’s are free to move but must remain on the inside Solutions are of the form: With nrg’s:
At T = 0, all states are filled up to the Fermi nrg A useful way to keep track of the states that are filled is: