Goldstein’s Experiment
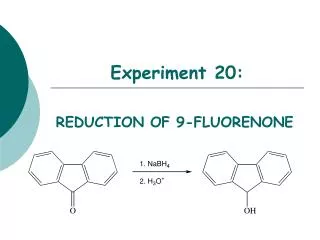
Goldstein’s Experiment. Discovered proton. -. H 2 gas. +. anode. perforated cathode. cathode rays. ZnS-coated fluorescent screen. elec. H 2 2 H 2 H 2 H + + 2 e -. elec. Thomson’s Model of Atom. + - + - + -.

Goldstein’s Experiment
E N D
Presentation Transcript
Goldstein’s Experiment • Discovered proton - H2 gas + anode perforated cathode cathode rays ZnS-coated fluorescent screen elec. H2 2 H 2 H 2 H+ + 2 e- elec.
Thomson’s Model of Atom + - + - + - + - + - + - + - + - + - + - + - + - + -
Fundamental Subatomic Particles • After Chadwick discovered the neutron in 1932, three fundamental subatomic particles had been identified: Relative Actual Relative Actual SymbolNameMassMassCharge Charge e-Electron 0 9.109 x 10-28 g -1 - 1.60 x 10-19 coul p+ Proton 1 1.673 x 10-24 g +1 + 1.60 x 10-19 coul n0 Neutron 1 1.675 x 10-24 g 0 0
Types of Radioactivity • In 1896, Henri Becquerel discovered radioactivity • In 1896, W. C. Roentgen discovered X-rays • Ernst Rutherford found 3 types of radioactivity + ß rays (particles) rays particles (rays) - slit
Rutherford’s Model of Atom Atom has a dense, hard nucleus surrounded by mostly empty space. e- = P+ 1. Approx. diam of atom. 10-10 m 2. Approx. diam. of nucleus 2 x 10-14 m
Moseley’s Experiment • vacuum + Anode - hot wire cathode high-energy e- Target metal Source of high-voltage electricity
Moseley’s Results Ti V Cr Mn Fe Co Ni Cu Zn • Energy of emitted X-rays = h = hc/ • Because wavelengths of emitted X-rays decreased across series of 1st-row transition elements, energies of emitted X-rays increased. • This meant that, for example, more energy was required to excite e- away from nucleus in V than in Ti • Therefore, nucleus of V is more positively charged than is nucleus of Ti • This led to CONECPT OF ATOMIC # decreasing
Mole - Mass Relationships of Elements Element Atom/Molecule Mass Mole Mass Number of Atoms 1 atom of H = 1.008 amu 1 mole of H = 1.008 g = 6.022 x 1023 atoms 1 atom of Fe = 55.85 amu 1 mole of Fe = 55.85 g = 6.022 x 1023 atoms 1 atom of S = 32.07 amu 1 mole of S = 32.07 g = 6.022 x 1023 atoms 1 atom of O = 16.00 amu 1 mole of O = 16.00 g = 6.022 x 1023 atoms 1 molecule of O2 = 32.00 amu 1 mole of O2 = 32.00 g = 6.022 x 1023 molecule 1 molecule of S8 = 2059.52 amu 1 mole of S8 = 2059.52 g = 6.022 x 1023 molecules
Molecular Mass - Molar Mass ( M ) The Molecular mass of a compound expressed in amu is numerically the same as the mass of one mole of the compound expressed in grams. For water: H2O Molecular mass = (2 x atomic mass of H ) + atomic mass of O = 2 ( 1.008 amu) + 16.00 amu = 18.02 amu Mass of one molecules of water = 18.02 amu Molar mass = ( 2 x atomic mass of H ) + atomic mass of O = 2 ( 1.008 g ) + 16.00 g = 18.02 g 18.02 g H2O = 6.022 x 1023 molecules of water = 1 mole H2O
One Mole of Common Substances CaCO3 100.09 g Oxygen 32.00 g Copper 63.55 g Water 18.02 g Fig. 3.2
Finding Moles • 1 mole = the amount of a substance that contains the same number of entities (formula units) as there are atoms in exactly 12.00 g of carbon-12. • 1 mol of carbon-12 contains 6.022 x 1023 atoms • 1 mol of H2O contains 6.022 x 1023 molecules • 1 mol of NaCl contains 6.022 x 1023 formula units of NaCl
Finding Moles • But, 1 mol of NaCl contains 6.022 x 1023 Na1+ ions and 6.022 x 1023 Cl1- ions, because: • NaCl Na1+ + Cl1- • 1 formula unit NaCl 1 Na1+ ion + 1 Cl1- ion or • 6.022 x 1023 formula units NaCl 6.022 x 1023 Na1+ ions + 6.022 x 1023 Cl1- ions or • 1 mol NaCl 1 mol Na1+ ions + 1 mol Cl1- ions so: • 1 mol Al(NO3)3 1 mol Al3+ ions + 3 mol NO31- ions
Information Contained in the Chemical Formula of Glucose C6H12O6 ( M = 180.16 g/mol) Carbon (C) Hydrogen (H) Oxygen (O) Atoms/molecule of compound Moles of atoms/ mole of compound Atoms/mole of compound Mass/moleculeof compound Mass/mole of compound 6 atoms 12 atoms 6 atoms 6 moles of 12 moles of 6 moles of atoms atoms atoms 6(6.022 x 1023) 12(6.022 x 1023) 6(6.022 x 1023) atoms atoms atoms 6(12.01 amu) 12(1.008 amu) 6(16.00 amu) =72.06 amu =12.10 amu =96.00 amu 72.06 g 12.10 g 96.00 g Table 3.2
Mass-Mole-Number Relationships • 1 mol atoms = 6.022 x 1023 atoms = molar mass (gAM) • 1 mol molecules = 6.022 x 1023 molecules = molar mass (gMM) • 1 mol formula units = ( ) x 6.022 x 1023 ions = molar mass (gFM) • Calculate # V atoms in 0.3 mol V • Method 1: 0.3 mol Vx atoms V 1 mol V 6.022 x 1023 V atoms x = 1.8066 x 1023 V atoms • Method 2: 0.3 mol V atoms x 6.022 x 1023 V atoms = 1.8066 x 1023 V atoms 1 mol V
Calculate the mass of 2 x 10-3 mol Rb atoms. • 2.00 x 10-3 mol Rb atoms x 85.47 g Rb = 1.71 x 10-1 g 1 mol Rb atoms • Calculate the # Cu atoms in 10 lb of Cu. • 10 lb Cu x 453.6 g Cu x 6.022 x 1023 atoms Cu = 4.3 x 1025 atoms 1 lb Cu 63.54 g Cu • Calculate the mol of CO2 in 50 g CO2. • 50.0 g CO2 x 1 mol CO2 = 1.14 mol CO2 44 g CO2 • So: # moles = mass/molar mass
Calculate the mass of 5 x 10-2 mol SO2. • Mass = # mol x molar mass • Mass = 5 x 10-2 mol x 64 g = 3.2 g SO2 1 mol • Calculate the # Cl1- ions in 3 x 102 g AlCl3. • 300 g AlCl3 x 6.022 x 1023 formula units AlCl3 x 3 Cl1- ions 133.33 g AlCl3 1 form.unit of AlCl3 • = 4.06 x 1024 Cl1- ions
Calculating Mass Percent • 1. From a formula • Mass % of element X = atoms of X in formula x atomic mass of X (amu) x 100 molecular (or formula) mass of compound (amu) or
Exact Stoichiometric Problem Calculate the kg of oxygen that are required to form 2.86 kg copper(I) oxide from reaction of oxygen with copper(I) sulfide, assuming complete reaction. • Step 1: Write balanced equation: copper(I) sulfide + oxygen copper(I) oxide + sulfur dioxide Cu2S(s) + O2(g) Cu2O(s) + SO2(g) Cu is balanced, sulfur is balanced, so balance oxygens: Cu2S(s) + 3 O2(g) 2 Cu2O(s) + 2 SO2(g) Now rebalance copper and sulfur: 2 Cu2S(s) + 3 O2(g) 2 Cu2O(s) + 2 SO2(g)
Step 2 Write theoretical mole ratios: 2 Cu2S(s) + 3 O2(g) 2 Cu2O(s) + 2 SO2(g) 3 mol 2 mol • Step 3 Write data given in problem: X kg 2.86 kg 2 Cu2S(s) + 3 O2(g) 2 Cu2O(s) + 2 SO2(g) 3 mol 2 mol • Step 4 Make necessary conversions: X kg x 103 g/kg 2.86 kg x 103 kg/g 2 Cu2S(s) + 3 O2(g) 2 Cu2O(s) + 2 SO2(g) 3 mol x 32 g/mol2 mol x 143.10 g/mol • Step 5 Set up proportion and solve: X x 103g=2.86 x 103g 3 x 32 g 2 x 143.10 g X = 2.86 x 103x 3 x 32 = 0.959 kg O2 2 x 143.10 x 103
Method 2 Solution • Step 1 Set up conversion problem using data given in problem 2.86 kg Cu2O x = ? kg O2 and stoichiometric factor (conversion factor) from theoretical mole ratios in the balanced equation: 2 Cu2S(s) + 3 O2(g) 2 Cu2O(s) + 2 SO2(g) so we have: 2.86 kg Cu2O x 103g x 1 mol Cu2O x 3 mol O2x 32 g O2 x 1 kg = 1 kg 143.10 g 2 mol Cu2O 1 mol O2 103 g • Step 2 Solve: = 0.959 kg O2
Types of Chemical Reactions • 1.Synthesis e.g. Mg + N2 ? Mg + N2 Mg2+ N3- Mg3N2 Element + Element Compound A + B AB
Balancing a Chemical Equation • 1. Begin with most complicated formula. • 2. Select element in that formula with highest-valued subscript. • 3. Count atoms of a given element on each side and make equal by algebra. If odd # on one side and even # on other side, use “crisscross” method to balance. e.g. unbalanced eq.: Mg + N2 Mg3N2 begin with Mg3N2 and balance Mg first: 3 Mg + N2 Mg3N2
More Synthesis Reactions • Complete and balance the following synthesis reactions: K + S8 Zn + P4 H2 + I2 N2 + H2 Answers: 16 K + S8 8 K2S 6 Zn + P4 2 Zn3P2 H2 + I2 2 HI N2 + 3 H2 2 NH3
Single Replacement Reactions Element + Compound Compound + Element