The Oxygen Cycle
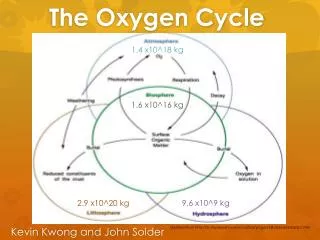
1.4 x10^18 kg. The Oxygen Cycle. 1.6 x10^16 kg. Kevin Kwong and John Solder. 2.9 x10^20 kg. 9. 6 x10 ^ 9 kg . Modified from http :// ilc.royalsaskmuseum.ca /ilc6/pages/62b/63e/pf63ep2p1.htm. Fluxes and Residence. Hydrosphere Calculation.


The Oxygen Cycle
E N D
Presentation Transcript
1.4 x10^18 kg The Oxygen Cycle 1.6 x10^16 kg Kevin Kwong and John Solder 2.9 x10^20 kg 9.6 x10^9kg Modified from http://ilc.royalsaskmuseum.ca/ilc6/pages/62b/63e/pf63ep2p1.htm
Hydrosphere Calculation Sea-Surface Dissolved Oxygen (0~600 m)5.4 x 10^16 mol Anoxic Zone (600~1100 m) Very Low DO Deep Ocean (1100~4000 m)2.08 x 10^20 mol X 32 g/mol => 6.65 x10^18 kg O2
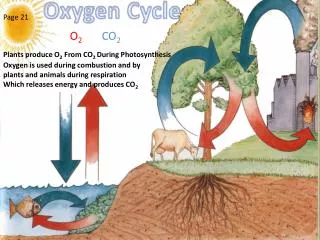
Photosynthesis and Respiration • 6CO2+ 6H2O + Energy → C6H12O6 + 6O2 • C6H12O6+ 6O2 →6 CO2+ 6H2O + Energy • 686 Kcal is the amount of energy imputed and released from these systems
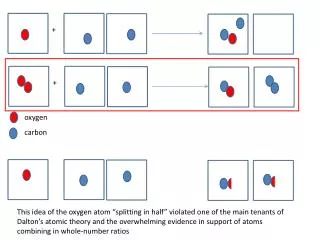
Photodissociation Weathering and Deposition • Oxidation of Earth Materials. 4FeO + O2 → 2Fe2O3 2 Ni + O2 = 2 NiO 2 Fe + SiO2 + O2 = Fe2SiO4 • Biological Weathering releases O2 from minerals as other nutrients are consumed. (Chelate compounds) • Corals and shells of marine organisms are O rich (CaCO3), becomes limestone. • Breakdown of Ozone by Photons. O3 + hν → O2 + O @ λ~240-310 nm O3 + O → 2 O2 2 O → O2 • The overall amount of ozone in the stratosphere is determined by a balance between production by solar radiation, and removal. The removal rate is slow, since the concentration of O atoms is very low.