Guidelines for Predicting Products for Chemical Equations
60 likes | 260 Vues
Guidelines for Predicting Products for Chemical Equations. http://www.ncpublicschools.org/docs/accountability/testing/eoc/Chemistry/chemistryreferencetable.pdf. Synthesis. Formation of binary compound: A + B → AB Metal oxide-water reactions: MO + H2O → base Base – M(OH)x

Guidelines for Predicting Products for Chemical Equations
E N D
Presentation Transcript
Guidelines for Predicting Products for Chemical Equations http://www.ncpublicschools.org/docs/accountability/testing/eoc/Chemistry/chemistryreferencetable.pdf
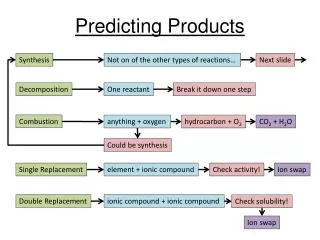
Synthesis • Formation of binary compound: A + B → AB • Metal oxide-water reactions: MO + H2O → base Base – M(OH)x • Nonmetal oxide-water reactions: (NM)O + H2O → acid Acid – H(NMO)
Decomposition • Binary compounds: AB → A + B • Metallic carbonates: MCO3 → MO + CO2 • Metallic hydrogen carbonates: MHCO3 → MO+ H2O(l) + CO2(g) • Metallic hydroxides: MOH → MO + H2O • Metallic chlorates: MClO3 → MCl + O2 • Oxyacidsdecompose to nonmetal oxides and water: acid → (NM)O + H2O
Single Displacement • Metal-metal replacement: A + BC → AC + B • Active metal replaces H from water: M + H2O → MOH + H2 • Active metal replaces H from acid: M + HX → MX + H2 • Halide-Halide replacement: D + BC → BD + C
Double Displacement • Formation of a precipitate from solution • Acid-Base neutralization reaction
Tips • Remember activity series, mainly for single displacement reactions • Apply solubility rules to ALLaqueous solutions • Write net ionic equations unless otherwise specified • H2CO3immediately becomes H2O and CO2 • H2SO3immediately becomes H2O and SO2 • NH4OHimmediately becomes H2O and NH3