Understanding Chemical Reactions: Predictions, Classifications, and Balancing
This guide provides a comprehensive overview of predicting products in single and double displacement reactions, along with acid-base interactions and combustion processes. Learn how a more active element can replace a less active one and the importance of solubility tables in determining the occurrence of reactions. Key examples showcase single displacement reactions, double displacement reactions, and neutralization reactions resulting in salt and water. Additionally, the guide offers practical exercises for predicting products and calculating solid product mass from reactants.

Understanding Chemical Reactions: Predictions, Classifications, and Balancing
E N D
Presentation Transcript
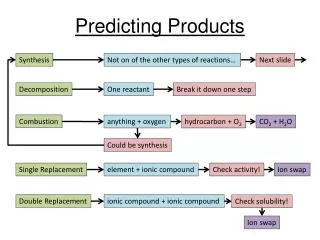
Single Displacement • Use activity series • A more active element replaces a less active one or the reaction will not occur
Double Displacement or Metathesis • Use solubility tables • For a reaction to occur, a ppt must form
NaCl(aq) + KBr(aq)----->NaBr(aq) + KCl(aq) No Reaction
Name reactants, predict & name products, and balance & classify the reactions on the board
DD or Metathesis • Acid-base: • When an acid is combined with a base, the products will be salt & water
Ba(OH)2(aq) + CaCl2(aq)ZnCl2(aq) + K2CrO4(aq)KCl(aq) + Na2SO4(aq)
Combustion • Carbon compound + oxygen yields Carbon dioxide & water
Predict Products & Balance Rxn • CaCl2 + F2 • CaCl2 + Na • CaCl2 + KNO3 • CaCl2 + K2SO4
Predict & Balance • AlBr3 + F2 • AlBr3 + Zn • AlBr3 + Na • AlBr3 + Ba(NO3)2 • AlBr3 + K2SO4
Calculate the mass of solid product formed when 4.16 g of barium chloride is added to 250 ml of 0.40 M potassium sulfate: