Understanding Acid-Base Concepts in Chemistry: Arrhenius, Bronsted, and Lewis Definitions
This lesson delves into the fundamental concepts of acids and bases as defined by Arrhenius, Bronsted, and Lewis. It covers key topics, including the definition of protons and hydronium ions, the release of hydrogen ions by mono-, di-, and tri-protic acids, and the mechanics of neutralization reactions. Additionally, it explores the Lewis definitions of acids and bases as electron pair acceptors and donors. Through examples and diagrams, students will grasp the relationships between conjugate acid-base pairs and their roles in various chemical reactions.

Understanding Acid-Base Concepts in Chemistry: Arrhenius, Bronsted, and Lewis Definitions
E N D
Presentation Transcript
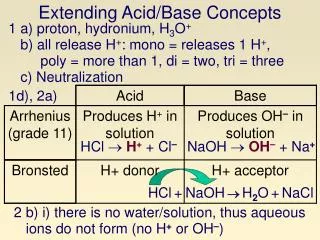
Acid Base Arrhenius (grade 11) Bronsted Extending Acid/Base Concepts 1 a) proton, hydronium, H3O+ b) all release H+: mono = releases 1 H+, poly = more than 1, di = two, tri = three c) Neutralization 1d), 2a) Produces H+ in solution Produces OH– in solution HCl H+ + Cl– NaOH OH– + Na+ H+ donor H+ acceptor HCl+NaOHH2O+NaCl 2 b) i) there is no water/solution, thus aqueous ions do not form (no H+ or OH–)
+ H H - H N + H Cl N H H + Cl H H Extending Acid/Base Concepts 2b) ii) diagram as on pg. 418 iii) HCl is an acid because it donates H+, NH3 accepts H+ and therefore is the base (NH4+ and Cl– then form an ionic compound) 3a) Lewis acid: electron pair acceptor Lewis base: electron pair donor 3b) the Lewis definition is universal because Arrhenius and Bronsted are special cases (as above or in reaction on pg. 428)
Extending Acid/Base Concepts 4 a) HCN(l) + H2O CN–(aq) + H3O+(aq) b) Forward: HCN is acid, H2O is base 4 a) HCN(l) + H2OCN–(aq) + H3O+(aq) c) Reverse: H3O+ is acid, CN– is base d) A conjugate acid-base pair are two substances that differ from each other by just one proton (H+) e) HCN and CN– and H2O and H3O+ are conjugate acid-base pairs See back of textbook for answers to PE 25 – 28, and RE 11.77 For more lessons, visit www.chalkbored.com