Recap – Ions and Ionic Bonding
This lecture covers the fundamental concepts of ionic and covalent bonding. It explains ions, their formation, and how cations and anions interact through electron transfer. The significance of energy in the formation of ionic bonds is highlighted, alongside the process of achieving noble gas electron configuration. Additionally, the principles of covalent bonding, including the sharing of electrons, are explored, showcasing examples like methane and water. Key learning outcomes include distinguishing between ionic and covalent bonds, depicting bonding diagrams, and understanding valence electrons.

Recap – Ions and Ionic Bonding
E N D
Presentation Transcript
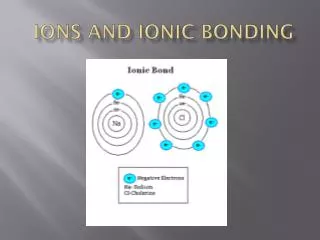
Recap – Ions and Ionic Bonding • Cations are positively charged and formed from metals. • Anions are negatively charged and formed from non-metals. • Cations and anions are formed at the same time by electron transfer. • Anionic bond is the electrostatic attraction between cations and anions.
Ionic Bonding Cl Na + • It takes energy to remove an electron from an atom. • Electrostatic attraction between cations and anions supplies energy. • Generally do not get very highly charges ions as too much energy is required to form them. - + Na Cl 6 230 kJ 1 090 kJ 2 360 kJ 4 630 kJ C C+ C2+ C3+ C4+ e- e- e- e-
F Sharing of Electrons In ionic compounds a Noble gas electron configuration is obtained by transfer of electrons. In covalent compounds a Noble gas electron configuration is obtained by sharing of electrons. F F F
Reason for Covalent Bonding • Electrons are shared to achieve a Noble gas configuration for each atom. • Electron density between the two atoms: the bond is directional. • Only electrons from the outermost shell can be involved in bonding – valence electrons. • Electrons tend to form pairs.
Covalent Bonding: C and H • Carbon has 4 electrons in outer shell – needs 8 • Hydrogen has 1 electron in outer shell – needs 2 H C H H H
Covalent Bonding - methane CH4 H C H H H
Covalent Bonding - water • Oxygen has 6 electrons in outer shell – needs 8 • Hydrogen has 1 electron in outer shell – needs 2 O H H
Covalent Bonding – H2O • We can identify bond pairs and lone pairs (or non-bonding pairs) of electrons O H H
Covalent Bonding: N and H • Show only valence electrons
Valence • Valence electrons are used in bonding. • The valence of an atom is the size of the charge if it forms an ion or the number of bonds it forms in a neutral molecule. 1 2 3 4 3 2 1
Diatomic Molecules • The diatomic elements (eg H2, N2, O2, F2) all have a similar formula but different bonding requirements. H2 Single Bond F2 Single Bond
Bonding Requirements • Double bonds occur where two electron pairs are shared between atoms. O2 Double Bond
Bonding Requirements • Triple bonds occur where three electron pairs are shared between atoms. N2 Triple Bond
Learning Outcomes: By the end of this lecture, you should: understand the difference between ionic and covalent bonding be able to draw diagrams representing the bonding in simple molecules know the difference between a bond pair and lone pair of electrons know the valence of elements in groups 1, 2, 13 – 17 be able to predict the formula of simple covalent compounds appreciate the difference between a single, double & triple bond be able to complete the worksheet (if you haven’t already done so…)
Questions to complete for next lecture: • Would the following pairs of elements form ionic or covalent bonds? H and O, Na and O, N and O, S and O. • Draw a diagram representing the molecule HCN. • How many lone pairs does each of the following molecules possess? H2, Cl2, H2S, HBr. • Oxygen has a valence of 2. Give an example of an ionic compound and a covalent compound containing oxygen that illustrates this concept. • Predict the formula of the simplest compounds formed between sulfur and fluorine, phosphorus and chlorine, magnesium and iodine.