Ionic Bonding
Ionic Bonding. Ionic Bonding. There are three types of chemical bonds, ionic , covalent , and metallic. The first type of bond we will study is the IONIC BOND.


Ionic Bonding
E N D
Presentation Transcript
Ionic Bonding • There are three types of chemical bonds, ionic, covalent, and metallic. • The first type of bond we will study is the IONIC BOND. • An ionic bond is an electrostatic attraction between a positive particle, also known as a CATION, and a negative particle, also known as an ANION.
Electron Dot Notation Before we can understand the formation of an ionic bond, we need to know two things: • The Octet Rule, one of the most important rules in chemistry. • How to write the Electron Dot Notation for each element on the Periodic Table.
1 2 3 6 5 8 4 7 Electron Dot Notation X represents the element symbol The dots represent valence electrons The pattern for drawing electrons helps us to understand how atoms lose or gain electrons. The maximum number of valence electrons is 8 .
The Octet Rule • The Octet Rule: Atoms will either lose, gain, or share valence electrons in order to obtain a full set of eight (8) valence electrons. • Valence- refers to the outer electrons in an atom. These are the electrons on the outer shell, which is the highest energy level.
Electron Dot Notation • For the Representative Groups, which are also called the A Groups, the Group Number is equal to the number of valence electrons. (The ones digit of group numbers 1 , 2 , through 18.) • Take out your Periodic Table and draw the electron dot notation for the first two elements in each group. The other elements in that group will be the same. >Use these example to help you draw dot notations. Be Li B C N O Ne F
Labeling the Periodic Table One exception: Helium only has two electrons, and we show them as a pair. Atoms which have a full set of valence electrons are very stable, as stable as Nobel Gases. (stable = not reactive) Skip the Transition Metals -
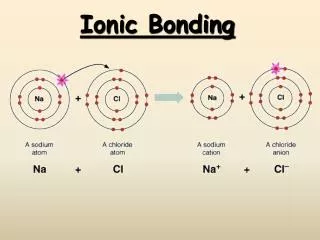
Formation of an Ionic Bond Ionic Bonds always involve the transfer of electrons from a metal to a nonmetal. They do this so that each may obey the Octet Rule.
Sodium Chloride (table salt) • The most famous “salt” of all! • Actually, any ionic compound is a salt! • The outer electron of the sodium transfers to the chloride • Then, the oppositely charged particles stick together by static electricity.
Metals form Cations Metals almost always lose their outer electrons, forming positive cations: Na Na+1 + 1 electron Ca Ca+2 + 2 electrons Al Al+3 + 3 electrons Each time, the metals lose their outermost valence electrons, and the energy level below is full.
Nonmetals form Anions Nonmetals almost always gain electrons, and become negative anions. Cl + 1e- Cl-1 Chlorine now has 18 e, just like Argon! O + 2e- O-2 N + 3e- N-3 Each time, the nonmetals gains the number of electrons needed to obtain 8 valence electrons.
Oxidation Numbers • An oxidation number is the charge an atom would have if it became an ion. • Take out your Periodic Table and label the A Groups as follows. AND Silver = +1 Zinc = +2 Cadmium = +2
Nomenclature • Nomenclature is a system for naming chemical compounds. • It has very specific rules you must follow. • We will start with the simplest type of ionic compound, a binary ionic compound. • Binary- means just two elements are present. • Ionic Bond--means a metal with a nonmetal
Naming Binary Ionic Compounds 1. Name the metal first 2. Then name the nonmetal, with an “ide” ending. • Examples: NaCl Sodiumchloride LiBr Lithium bromide CaF2 Calcium fluoride
Formula Writing for Binary Ionic Compounds • Sometimes you will be given the name of a compound and be asked to write the chemicalformula. • This requires three careful steps, and a periodic table. • Do not guess. Follow the steps! • “Almost right” is wrong!
Formula Writing Example: Aluminum Oxide STEP 1: write the symbols, metal first, then nonmetal. Al O STEP 2: above each symbol, write the oxidation number Al+3 O-2 STEP 3: Add subscripts so that the overall charge of the compound is zero. (“criss-cross” + 6 and - 6 = 0) Step 4:(Erase the oxidation numbers, they are NOT part of the formula, they just help us get the correct subscripts!) Al2O3 +3 x 2 = +6 -2 x 3 = -6 subscripts
Transition Metals – Groups 3 – 12 The B Groups • Many transition metals have more than one possible oxidation state. (Sometimes they give away one electron, sometimes two, etc. up to losing 7 electrons! ) • Silver is always +1, Zinc is always +2, and Cadmium is +2 • Some other metals like Lead and Tin, also require a Roman Numeral in naming compounds
Which elements need a roman numeral?It’s faster to say which ones don’t!(There are a few others, but we won’t ask about them—okay?) Mark these on your periodic table.
Roman Numerals Examples: NameFormula Copper (I) Cu+1 Iron (II) Fe+2 Chromium (III) Cr+3 Tin (IV) Sn+4 It is helpful to label your periodic table with the oxidation states of the common ones.
Naming Transition Metal Compounds(except zinc, cadmium—always +2, and silver always +1…) If you notice that the formula of a compound has a transition metal, (except Zn, Cd, Ag) you must use a Roman numeral in the compound name. Examples: FeCl2 Is Iron (II) chloride CuO Is Copper (II) oxide Cr3N2 Is Chromium (II) nitride Notice that the Roman numeral corresponds to the subscript of the OTHER ATOM.
Writing Transition Metal Formulas(except zinc, cadmium—always +2, and silver always +1…) Copper (II) fluoride Step 1 Write element symbols CuF Step 2 Put oxidation numbers above each element (The Roman numeral “(II)” gives the oxidation number for copper as +2). Cu+2 F-1 Step 3Add subscripts tomake the charge total = zero, i.e. “criss-cross”. Note: Use parentheses if you need more than one of a polyatomic ion on other formulas. CuF2 Copper II phosphate Cu+2 PO4-3 --- > Cu3(PO4)2
Practice Together Write formulas for the following: Tin (II) chloride Copper (I) oxide Lead (IV) sulfide Name the following compounds: PbCl2 CuO SnCl4
Polyatomic Ions • Many common chemical formulas contain polyatomic ions, which are groups of atoms bonded together, that carry a charge. • See your list of polyatomic ions ON your class periodic table. • Polyatomic ions stay together as a group as they undergo reactions.
Common Polyatomic Ions Hydroxide OH-1 Nitrate NO3-1 Sulfate SO4-2 Chlorate ClO3-1 Ammonium NH4+1 Mostpolyatomic ions are negative, meaning they have extra electrons. Ammonium is the only polyatomic ion we use that is positive. It has given away an electron.
Compounds with Polyatomic Ions These compounds will have at least three elements, a metal, and then a polyatomic ion. Name the metal first, and then the polyatomic ion. Don’t change the name of the polyatomic ion!
Naming Ternary Compounds (3 or more elements) NaNO3 sodium nitrate CaSO4 calcium sulfate Ba(OH)2 barium hydroxide NH4Cl ammonium chloride KClO3 potassium chlorate When you see three or moreelements, look for a hidden polyatomic ion ! Never say “calcium sulfur oxide.”
Writing Formulas for Compounds Containing Polyatomic Ions Get out a periodic table and a formula chart with the polyatomic ions on it. Use the three step process. Example: Calcium chlorate Step 1 Write the symbols (polyatomic ion formula representing the name given) CaClO3 Step 2 Label chargesCa+2 ClO3-1 Step 3 Write subscripts that balance the charges Ca(ClO3)2 Step 4: Erase charges from your formula
Properties of Ionic Compounds • Many ionic compounds dissolve in water • Solutions of ionic compounds conduct electricity. Ions can move! • These solutions are called “Electrolytes” • Ionic compounds have very high melting points • When melted “molten” they conduct electricity. Ions can move! (melted salts are conductors) • Exist as crystals in a “crystal lattice.”
Acids • The acids that we will study in this unit are easy to recognize, they contain one or more hydrogen atoms at the beginning of their formulas. • An acid is a compound that dissociates (splits up) in water to produce hydrogen ions. • Example: Hydrochloric acid ---breaks apart in water to form -- > hydrogen ions and chloride ions. HCl H+1 + Cl-1
Binary Acid Nomenclature • Binary means two elements. • These will have hydrogen and one other element. 1. Use the prefix Hydro- 2. Put an –ic ending on the second element 3. Add the word “acid” HCl Hydrochloric acid HBr Hydrobromic acid HF Hydrofluoric acid
Naming Oxyacids • These acids will contain hydrogen and a polyatomic ion which has one or more oxygens • If the ion ends in –ate change it to an –ic ending HNO3 Nitric acid (Contains the nitrate ion) H2SO4 Sulfuric Acid (Contains the sulfate ion) “Yuck! I ATE something ICKY!”
Naming Oxyacids • If the polyatomic ion ends in –ite, change it to –ous. • Add the word “acid” • Ex: HNO2 Nitrous acid Nitrite ion H2SO3 Sulfurous acid Sulfite ion Bites are delicious