Ionic bonding
Ionic bonding. Along with nomenclature of ionic compounds, acids, and molecular compounds. Terms. Formula unit – ionic substances, smallest whole # ratio Molecular formula – covalent compounds, actual # of atoms in one molecule


Ionic bonding
E N D
Presentation Transcript
Ionic bonding Along with nomenclature of ionic compounds, acids, and molecular compounds
Terms • Formula unit – ionic substances, smallest whole # ratio • Molecular formula – covalent compounds, actual # of atoms in one molecule • Empirical formula – smallest whole # ratio (ionic formulas are empirical) • Hydrates – # water molecules attached to each molecule • Anhydrous – water has been removed • Hygroscopic – attracts water • Deliquescent – attracts enough water to dissolve
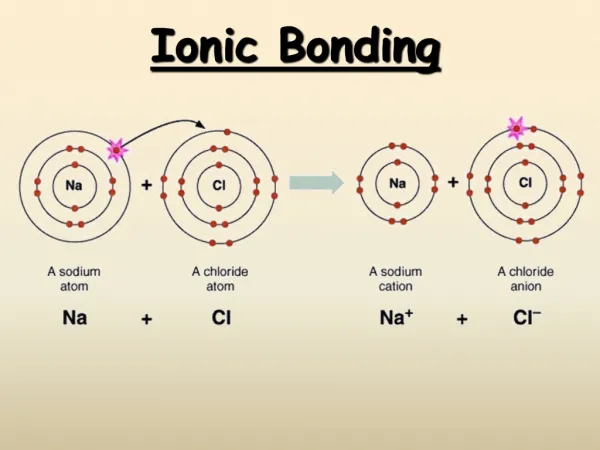
Ionic Bonds • TRANSFER of electrons • Cation + anion • Metals lose their valence e- and become positively charged • Nonmetals gain enough e- to have a full outer shell (octet, or 8) and become negatively charged • Opposites attract! Ionic compounds are formed • Electrostatic forces hold ions together
Properties of Ionic Bonding • Hard, brittle, crystalline solids at room temperature • High melting points • DO NOT conduct as solids • DO conduct when melted or dissolved in water • Most are soluble in water • Link
Formulas • Chemical formula • Shows kinds and numbers of atoms in smallest representative unit • NaCl, H2O • Formula unit • Ionic compounds form CRYSTALS that vary in size, because the ions attract from all sides, the ions pack together in an alternating pattern, so the formula shows the lowest whole number ratio of ions
Writing ionic formulas • There are some steps to writing ionic formulas: • The cation always goes first • The anion goes second • The charge of cation becomes the subscript for the anion • The charge of the anion becomes the subscript for the cation • The subscript tells you how many of that cation/anion is needed to bond to the cation/anion
PracticeK + F Al + O Be + S Al + BrK + I Mg + I Na + O • Sometimes one loses more e- than the other needs, or vice versa, so the ratio cannot be 1:1 • look at the charges, criss-cross them so that they become the subscripts, then reduce if possible; the idea is that the overall charge in a chemical formula is ZERO • Al+3 O-2 Al2O3 • Be+2 S-2 Be2S2 BeS 2:2 reduces to 1:1
Mark your Periodic Table! All the transition metals except Ag and Zn can have more than one charge and need a Roman numeral (Pb and Sn need one too!) +2 +1 +2 +4 +2 +4
Metallic Bonding • Cations in a sea of electrons • Only occurs in pure metal elements and mixtures • Properties include • Malleable, can be flattened into thin sheets • Ductile, can be pulled into wire • Bendable/shapeable vs brittle ionic compounds • Alloys – metal mixtures, designed for specific qualities • Durability, strength, resistance to corrosion, lightweight, etc.
Covalent Bonding • sharing of electrons to form molecules • Properties of covalent compounds: • lower melting and boiling points than ionic compounds • soft and squishy • more flammable than ionic compounds • don't conduct electricity in water • Are not soluble in water • Coordinate covalent bonding occurs when one element contributes both electrons of the electron pair when the chemical bond forms
Network Solids • Network Covalent: this is a special type of nonpolar covalent bonding • Rather than forming discrete molecules, the atoms bond in a continuous, 3-D fashion forming crystalline solids, much like ionic solids– but with some significantly different properties • Examples: sand (SiO2), diamond (carbon), graphite (carbon) • high melting point • NOT SOLUBLE • NOT CONDUCTIVE
Naming Ionic Compounds • Remember, ionic compounds almost always have a metal • Cations have the same name as the metal (add “ion”) • Anions have the ending changed to –ide, or they are polyatomic and have a special name • Metals that can form more than one type of ion use a Roman numeral equal to the charge to designate the type • Fe+2 is iron(II) • Fe+3 is iron(III)
Naming Covalent Compounds • Use prefixes to indicate the # of atoms of each element, also use for hydrates (•H2O) • Mono, di, tri, tetra, penta, hexa, hepta, octa, nona, deca; don’t use mono for 1st element • Change ending to –ide • NO2 nitrogen dioxide • N2O4 dinitrogen tetroxide (drop the a from the prefix when followed by o) • CuSO4•5H2O copper(II) sulfate pentahydrate
Organic Compounds • Organic – contains carbon and hydrogen • These have their own special naming system, using different prefixes for the # of carbons, and suffixes indicating the types of bonds
1st indicate # of Carbons 1-meth 2-eth 3-prop 4-but 5-10 same as inorganic prefixes Then indicate type of bonds All single bonds then tack on -ane CnH2n+2 A double bond add -ene A triple bond add -yne Methane, propene, butyne Naming Organic Compounds
Naming Acids • Binary Acids (HCl) • “hydro” prefix • Root of anion (2nd element) • “ic” suffix • + acid • HCl is hydrochloric acid • HF hydrofluoric acid
Naming Acids - polyatomic • MUST recognize the polyatomic ion • Change “ite” to “ous” • Change “ate” to “ic” • + acid • H3PO4: • (PO4)- phosphate phosphoric acid • All negative polyatomic ions can be acids, just put hydrogen in front of formula (subscript will be charge of ion)