Ionic Bonding
Ionic Bonding. Few elements exist in a pure form. Most exist in combination with other element(s) The force that holds two atoms together is known as a chemical bond. Three types of chemical bonds: Ionic Covalent Metallic. Ionic Compounds.


Ionic Bonding
E N D
Presentation Transcript
Few elements exist in a pure form. • Most exist in combination with other element(s) • The force that holds two atoms together is known as a chemical bond. • Three types of chemical bonds: • Ionic • Covalent • Metallic
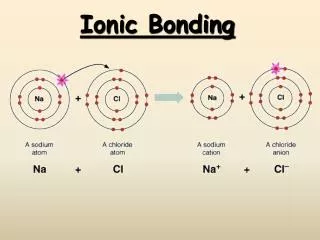
Ionic Compounds • Ionic bonding involves the transfer of electrons from one neutral atom to another • Cation:positively charged ion (lose electrons) • Anion: negatively charged ion (gain electrons) • Ionic compounds form from two or more ionsbeing held together by electrostatic forces.
Ionic Compounds • Ionic compounds form between two atoms with large differences in electronegativity, generally greater than 1.67
The periodic table helps us predict what • type of ion an element will form • Will an element gain or lose electrons? • Number of valence electrons • How close to noble gas status
Metals:tend to lose electrons and become cations Nonmetals:tend to gain electrons and become anions Ionic bonds occur betweenmetals andnonmetals
Ionic Bonding Na Cl
Ionic Bonding Na+ Cl-
The overall charge of a formula unit for an ionic compound is zero. • The charges of the ions cancel each other out. • Atoms combine in multiples so that the overall charge of the compound is zero.
Na +1 + Cl -1 = NaCl Na +1 + S -2 = Na2S Ca +2 + Cl -1 = CaCl2 Ca +2 + P -3 = Ca3P2 The charge of the ion is referred to as its oxidation number
Ionic Compounds A compound that is composed of ions is called an ionic compound. Note that only the arrangement of electrons has changed. Nothing about the atom’s nucleus has changed.
Properties of Ionic Compounds Ionic compounds have a crystalline structure, a regular repeating arrangement of ions in the solid. Even though the ions are strongly bonded to one another, ionic compounds are brittle.
Repeating arrangement of ions in the solid form a crystalline structure
Properties of Ionic Compounds Rigid Brittle High melting points because of strong forces between ions. Conduct electricity in the molten and dissolved states only (form electrolytes)
Lattice Energy The energy required to separate one mole of the ions of an ionic compound is called lattice energy, which is expressed as a negative quantity. The greater (the more negative) the lattice energy, the stronger the force of attraction between the ions.
Lattice Energy Lattice energy tends to be greater for more highly-charged ions (those atoms that have more electrons to give or those atoms that can take more electrons). Lattice energy also tends to be greater for small ions.
Question NaCl or MgS (MgS would have the higher lattice energy because Mg can give away more electrons than Na and sulfur can take more electrons than Cl.) Between the following ionic compounds, which would be expected to have the higher (more negative) lattice energy?
Question LiF or KBr (LiF would have the higher lattice energy because Li is smaller than K and F is smaller than Br.) Between the following ionic compounds, which would be expected to have the higher (more negative) lattice energy?
Ionic Compounds Binary compounds: ionic compounds consisting of two elements. Ternary compounds: ionic compounds composed of a metal and a polyatomic ion Polyatomic Ion: a group of bonded atoms that are treated as one unit and carry and overall charge
Naming Binary Ionic Compounds When naming and writing formulas, metal (cation) is first and stated as the element name Na +1 = sodium The nonmetal (anion) is named next, with the element name being changed to -ide Cl-1 = chloride ion NaCl =sodium chloride
Naming Binary Ionic Compounds Transition metals may form cations with different charges The charge of the cation is indicated in Roman numerals after the name of the metal. It is NOT included in the formula. Iron (II) oxide = FeO
Naming Ternary Ionic Compounds Polyatomic ions contain at least two elements and carry an overall charge All but one polyatomic ion have a negative charge (anion). Lists of common polyatomic ions can be found on the Chem. Reference Table and in textbook