Understanding Ionic Bonding and its Role in Classifying Pure Substances
This resource delves into ionic bonding, which involves the attraction between positive and negative ions forming ionic compounds, typically in a crystal lattice structure. It covers how ionic compounds differ from molecular compounds, the rules of electron transfer, and the formation of common ionic compounds such as sodium chloride and magnesium sulfide. The document also explains how to predict formulas for ionic compounds, detailing the role of polyatomic ions and providing examples of charge cancellations. Perfect for enhancing understanding of ionic bonding in chemistry.


Understanding Ionic Bonding and its Role in Classifying Pure Substances
E N D
Presentation Transcript
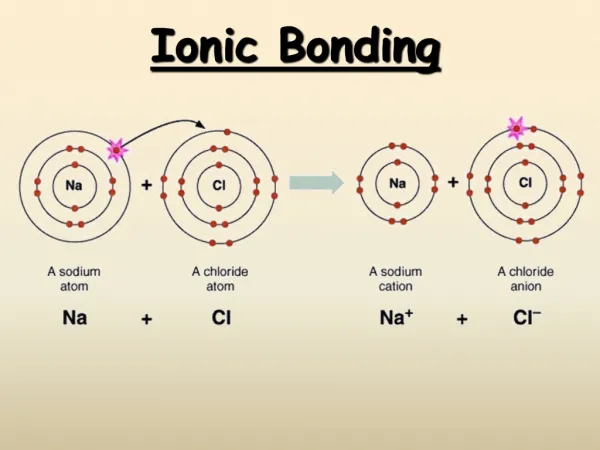
Ionic Bonding Mr. Thomson Experience Academy
Classifying Pure Substances http://wps.prenhall.com/wps/media/objects/476/488316/Instructor_Resources/Chapter_05/FG05_07.JPG
Comparison – Bond Types http://www.elmhurst.edu/~chm/vchembook/images/150compare.GIF
Ionic Bonding • Ionic Bond: Attraction between positive and negative ions that holds atoms in place in an ionic compound (usually a crystal). • Ionic compounds usually form when a metal positive ion (cation) bonds with a non-metal negative ion (anion). • Ionic compounds involve the transfer of electrons. (Unlike covalent bonding, where electrons are shared.)
Crystal Structure of Ionic Compounds • Ionic compounds do not form molecules. Instead, ionic compounds form an organized three-dimensional structure of alternating positive and negative ions. http://www.mtl-system.com/Web%20Images/Model_chemistry/Large/Molecular%20Model/T22003_l.jpg
Comparison: Molecules and Ionic Compounds http://www.physchem.co.za/Bonding/Molecule.htm Water, a molecular compound, is made of billions and billions of INDIVIDUAL water molecules. Salt, an ionic compound, is made of billions and billions of ions, all together in a crystal lattice. There are NO individual molecules of NaCl. http://wps.prenhall.com/wps/media/objects/476/488316/Instructor_Resources/Chapter_12/FG12_02.JPG
How are ions formed? • When atoms form ions, they lose or gain electrons in order to have eight valence electrons.
How is sodium chloride formed? Animation(Source: http://www.bios.niu.edu/sims/chemistry/Chemistry3.htm)
Na Cl Na 1s22s22p63s1 Remember the “rule of eight.” Sodium wants a full outer (valence) shell. So sodium can either gain seven electrons to fill the third shell, or get rid of the 3s1 electron. Then the outer shell (second) will be full. The new electron configuration will be: Na+ 1s22s22p6
Na Cl Na+ 1s22s22p6 Why is sodium +1 when it loses an electron? Na Na+ p+ = 11 p+ = 11 e- = 11 e- = 10 Zero charge +1 charge
Na Cl Cl 1s22s22p63s23p5 Remember the “rule of eight” again. Chlorine wants a full shell also. So it can either lose seven electrons, or gain one electron. The new electron configuration will be: Cl - 1s22s22p63s23p6
Na Cl Cl - 1s22s22p63s23p6 So why is chlorine -1 when it gains an electron? Cl Cl - p+ = 17 p+ = 17 e- = 17 e- = 18 Zero charge -1 charge
Sodium Chloride Na Cl -1 +1 Na Cl
Crystal Structure of Sodium Chloride Source: http://www.geog.ouc.bc.ca/physgeog/contents/images/sodiumchloride.jpg
Another Look at Sodium Chloride Crystal http://library.tedankara.k12.tr/chemistry/vol1/salts/trans19.htm
Na Cl In all ionic compounds, the charges cancel, so the overall charge for the compound is zero.
Magnesium sulfide Mg S [Ne]3s2 [Ne]3s23p4 Mg S
Magnesium sulfide Mg S [Ne]3s2 [Ne]3s23p4 Mg S
Magnesium sulfide Mg S [Ne]3s2 [Ne]3s23p4 -2 +2 Mg S Extra Electrons from Magnesium Mg +2 S -2 [Ne] [Ne]3s23p6
Magnesium sulfide Mg +2 S -2 [Ne] [Ne]3s23p6 MgS
Calcium chloride Ca +2 Cl -1 CaCl2 When the charges don’t cancel…cross the charges to write the correct formula.
Aluminum Oxide Al +3 O -2 Al2O3 When the charges don’t cancel…cross the charges to write the correct formula.
Polyatomic Ions Polyatomic ions: a charged group of covalently bonded atoms. The charge comes from gaining or losing electrons as a group. (See back of your periodic table.) • Examples: • Ammonium: NH4+ • Nitrate: NO3-
Sodium nitrate Na +1 NO3-1 Do the charges cancel? Yes…so just write the formula. NaNO3
Potassium carbonate K +1 CO3-2 Do the charges cancel? No…so cross the charges. Answer: K2CO3
Ammonium sulfate NH4+1 SO4-2 Charges cancel? No…so cross the charges. (NH4)2SO4
Calcium Phosphate Ca +2 PO4-3 Charges cancel? No…so cross the charges. Ca3(PO4)2
Classwork Homework: Naming Ionic Compounds WS 1 Ionic Bonding Classwork Both Stamped Tomorrow Many of you still need to finish the Molecule Building Activity…this will need to be completed on your own time… Many of you also still need to turn in your Online Bonding Activity.