Understanding Ionic Bonding: Principles and Properties
Learn about ionic bonds between metals and non-metals, valence electrons, coordination numbers, and formation of ionic compounds. Understand how cations and anions interact to create stable compounds. Discover the characteristics of ionic compounds and their properties.


Understanding Ionic Bonding: Principles and Properties
E N D
Presentation Transcript
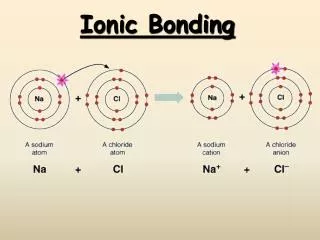
Ionic bonds are characterized as: - bonds between metals & non-metals metals tend to _____ electrons non-metals tend to _____electrons - bonds between cations & anions cations are _______ anions are _______ - bonds involving a transfer of e-(from a metal to a nonmetal) - Ionic bonds are also termed Salts
Valence Electrons: - What are Valence Electrons? How do we know how many there are? -What rule must be meet for a stable compound?
Ionic compounds form crystalline lattice structures which are very strong. High Melting Point Ex. Melting point for NaCl is 800°C
Coordination Number: Coordination Numbers: the # of ions of opposite charge surrounding it The number of ions of opposite charge surrounding it Both the sodium and chlorine have 6 NaCl Both the cesium and chlorine have 8 CsCl Each titanium has 6, and each oxygen has 3 TiO2
Starting Valence e- = 8 Final Valence e- = 8 End Total Charge = 0 [Ne]3s1 [Ne]3s23p5 [Ne] [Ne]3s23p6 Na Cl Na+ Cl - + + Cl Starting Total Charge = 0 - Na Na+ Cl Cations have smaller atomic radii Anions have larger atomic radii Remember!!!
When ionic compounds dissolve into solution, their + and – ions separate (electrolyte). + = Na+(aq) - = Cl-(aq) H2O surrounds ions in solution to prevent cations & anions from recombining (solvation)
Good electrolytes, like many ionic compounds are good conductors of electricity. Electrolysis
How do ionic compounds with subscripts form? Ex. How does MgCl2 form? Cl Cl - Cl Mg 2+ - Mg Cl
Diagram the formation of the ionic compound AlF3! - F F F F F F Al - 3+ Al -
Find the balanced ionic formula for Calcium & Bromine. Br Br Br Br - Ca 2+ Ca - CaBr2 Ca oxidation # = +2 We need a 2nd Br atom to balance the net charge! Br oxidation # = -1