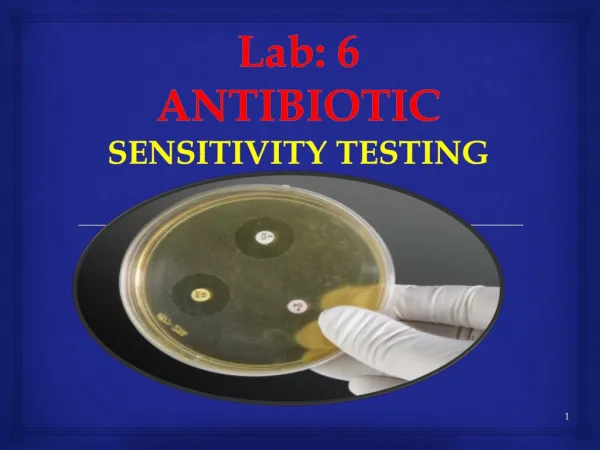
ANTIBIOTIC SENSITIVITY TESTING SKILL BASED LEARNING
ANTIBIOTIC SENSITIVITY TESTING SKILL BASED LEARNING. Dr.T.V.Rao MD. Uses of Antibiotic Sensitivity Testing. Antibiotic sensitivity test : A laboratory test which determines how effective antibiotic therapy is against a bacterial infections.


ANTIBIOTIC SENSITIVITY TESTING SKILL BASED LEARNING
E N D
Presentation Transcript
ANTIBIOTIC SENSITIVITY TESTINGSKILL BASED LEARNING Dr.T.V.Rao MD Dr.T.V.Rao MD
Uses of Antibiotic Sensitivity Testing • Antibiotic sensitivity test: A laboratory test which determines how effective antibiotic therapy is against a bacterial infections. • Antibiotic sensitivity testing will control the use of Antibiotics in clinical practice • Testing will assist the clinicians in the choice of drugs for the treatment of infections. Dr.T.V.Rao MD
What is the goal of Antibiotic Sensitivity testing? • The goal of antimicrobial susceptibility testing is to predict the in vivo success or failure of antibiotic therapy. Tests are performed in vitro, and measure the growth response of an isolated organism to a particular drug or drugs. The tests are performed under standardized conditions so that the results are reproducible. The test results should be used to guide antibiotic choice. The results of antimicrobial susceptibility testing should be combined with clinical information and experience when selecting the most appropriate antibiotic for our patients. Dr.T.V.Rao MD
Components of Antibiotic Sensitivity Testing • 1.The identification of relevant pathogens in exudates and body fluids collected from patients • 2. Sensitivity tests done to determine the degree of sensitivity or resistance of pathogens isolated from patient to an appropriate range of antimicrobial drugs • 3. Assay of the concentration of an administered drug in the blood or body fluid of patient required to control the schedule of dosage. Dr.T.V.Rao MD
Why Need Continues for Testing Antibiotic Sensitivity • Bacteria have the ability to develop resistance following repeated or subclinical (insufficient) doses, so more advanced antibiotics and synthetic antimicrobials are continually required to overcome them. • Antibiotic sensitivity testing is essential part of Medical Care Dr.T.V.Rao MD
Introduction • Susceptibility test, main purposes: • As a guide for treatment • Sensitivity of a given m.o. to known conc. of drugs • Its concentration in body fluids or tissues • As an epidemiological tool • The emergence of resistant strains of major pathogens (e. g. Shigella, Salmonella typhi) • Continued surveillance of the susceptibility pattern of the prevalent strains (e. g. Staphylococci, Gram-negative bacilli) Dr.T.V.Rao MD
Introduction • Methods for antimicrobial susceptibility testing • Indirect method • cultured plate from pure culture • Direct method • Pathological specimen • e.g. urine, a positive blood culture, or a swab of pus Dr.T.V.Rao MD
What Does the Laboratory Need to Knowabout Antimicrobial Susceptibility Testing (AST) ? • Which organisms to test? • What methods to use? • What antibiotics to test? • How to report results? Dr.T.V.Rao MD
Routine Susceptibility Tests • Disk diffusion (Kirby Bauer) • Broth micro-dilution MIC • NCCLS reference method • Etest Dr.T.V.Rao MD
Preparing for Testing • Inoculum preparation - Number of test organisms can be determined using different methods: • Direct count (Microscopic examination) • The optical density (OD) at 600 nm (Spectrophotometry) • Plate count: making dilution first • Turbidity standard (McFarland) routinely performed. Dr.T.V.Rao MD
Choosing the Appropriate Antibiotic • Drugs for routine susceptibility tests: • Set 1:the drugs that are availablein most hospitals and for which routine testing should be carried out for every strain • Set 2:the drugs that are tested only: • at the special request of the physician • or when the causative organism isresistantto the first-choice drugs • or when other reasons (allergy to a drug, or its unavailability) make further testing justified Dr.T.V.Rao MD
Table 1: Basic sets of drugs for routine susceptibility tests(http://w3.whosea.org/) Dr.T.V.Rao MD
Antimicrobial Susceptibility Testing • Diffusion method • Put a filter disc, or a porous cup/a bottomless cylinder containing measured quantity of drugs on the a solid medium that has been seeded with test bacteria • Dilution method • vary amount of antimicrobial substances incorporated into liquid or solid media • followed by inoculation of test bacteria Dr.T.V.Rao MD
Susceptibility Testing Methods Incubate plate 18-24 hr, 35 C Measure and record zone of inhibition around each disk Inoculate MH plate Place disks on agar plate
Diffusion Method • Disc diffusion method : The Kirby-Bauer test • Antibiotic-impregnated filter disc* • Susceptibility test against more than one antibiotics by measuring size of “inhibition zone ” • 1949: Bondi and colleagues paper disks • 1966: Kirby, Bauer, Sherris, and Tuck filter paper disks • Demonstrated that the qualitative results of filter disk diffusion assay correlated well with quantitative results from MIC tests Dr.T.V.Rao MD
Disc Diffusion Method • Procedure(Modified Kirby-Bauer method: National Committee for Clinical Laboratory Standards. NCCLS) • Prepareapproximately.108 CFU/ml bacterial inoculum in a saline or tryptic soy broth tube(TSB) or Mueller-Hinton broth (5ml) • Pick 3-5 isolated colonies from plate • Adjust the turbidity tothesame as the McFarland No. 0.5 standard.* • Streak the swabon the surface of the Mueller-Hinton agar(3 times in 3 quadrants) • Leave 5-10 min to dry the surface of agar Dr.T.V.Rao MD
Examining purity of plateSelect the Colonies from Pure Isolates Transmitted light Reflected light Dr.T.V.Rao MD
Disk Diffusion Test Prepare inoculum suspension Prepare inoculum suspension Select colonies Dr.T.V.Rao MD
Prepare the Material for Inoculation Standardize inoculum Suspension as per Mac farland standard Mix well Dr.T.V.Rao MD
Swab the plate with optimal sample Remove sample Swab plate Dr.T.V.Rao MD
Select the Disks and Apply Select disks Dr.T.V.Rao MD
Incubate Overnight Dr.T.V.Rao MD
Disc Diffusion Method • Place the appropriate drug-impregnated disc on the surface of the inoculated agar plate • Invert the plates and incubate them at35oC, o/n (18-24 h) • Measure the diameters of inhibition zone in mm Dr.T.V.Rao MD
Read the Results with Precision Transmitted Light Dr.T.V.Rao MD
Disc Diffusion Method • Measurement of the diameters of inhibition zone • Measure from the edge where the growth stats, BUT there are three exceptions • With sulfonamides and co-trimoxazole, ignore slight growth within the zone • Certain Proteus spp. may swarm into the area of inhibition • When beta-lactamase producing Streptococci are tested, zone of inhibition are produced with a heaped-up, clearly defined edge, regardless of the size of the inhibition zone, they should be reported as resistant Dr.T.V.Rao MD
Look at the Charts for establishing the zones of Sensitivity • The zone sizes are looked up on a standardized chart to give a result of sensitive, resistant, or intermediate. Many charts have a corresponding column that also gives the MIC (minimal inhibitory concentration) for that drug. Dr.T.V.Rao MD
Disc Diffusion Method Reporting the Results • Interpretation of results • By comparing with the diameters with “standard tables” • Susceptible • Intermediate susceptible • Low toxic antibiotics: Moderate susceptible • High toxic antibiotics: buffer zone btw resistant and susceptible • Resistant Dr.T.V.Rao MD
Factors Affecting Size of Zone of Inhibition • Larger zones with light inoculum and vice versa • If after application of disc, the plate is kept for longer time at room temperature, small zones may form • Larger zones are seen with temperatures < 35oC • Ideal 16-18 hours; less time does not give reliable results • Inoculum density • Timing of disc application • Temperature of incubation • Incubation time Dr.T.V.Rao MD
Factors Affecting Size of Zone of Inhibition • Size of the plate • Depth of the agar medium(4 mm) • Proper spacing of the discs (2.5 cm) • Smaller plates accommodate less number of discs • Thin media yield excessively large inhibition zones and vice versa • Avoids overlapping of zones Dr.T.V.Rao MD
Factors Affecting Size of Zone of Inhibition • Deterioration in contents leads to reduced size • Affects rate of growth, diffusion of antibiotics and activity of antibiotics • Tetracycline, novobiocin, methicillin zones are larger • Aminoglycosides, erythromycin zones are larger • Subjective errors in determining the clear edge • Potency of antibiotic discs • Composition of medium • Acidic pH of medium • Alkaline pH of medium • Reading of zones Dr.T.V.Rao MD
Quality Assurance in Antibiotic Susceptibility Testing • Visit - WHO-Regional Office for South East Asia website • Medium:Mueller-Hinton agar plates • Enterococcus faecalis (ATCC 29212 or 33l86) and a disc of co-trimoxazole 20 mm in diameter of the inhibition zone • Procedure: Modified Kirby-Bauer method recommended by National Committee on Clinical Laboratory Services (NCCLS) • Susceptibility test with quality control strains Dr.T.V.Rao MD
Quality Assurance in Antibiotic Susceptibility Testing with Control strains • Susceptibility test with quality control strains for every new batch of Mueller-Hinton agar • Staphylococcus aureus (ATCC 25923) • Escherichia coli (ATCC 25922) • Pseudomonas aeruginosa (ATCC 2785 ) Dr.T.V.Rao MD
Quality Assurance in Antibiotic Susceptibility Test • Salient features of quality control • Use antibiotic discs of 6 mm diameter • Use correct content of antimicrobial agent per disc • Store supply of antimicrobial discs at -20oC • Use Mueller-Hinton medium for antibiotic sensitivity determination • Use appropriate control cultures • Use standard methodology for the test Dr.T.V.Rao MD
Need for Modified Methods • Modified Methods in Disc diffusion for Antibiotic sensitivity testing to be used for detections of following bacterial isolates • 1 MRSA • 2 ESBL • 3 Enterobacteriaceae and Gram negative bacteria and Carbapenems resistant using Modified Hodge test Dr.T.V.Rao MD
Dilution Method • Minimum Inhibition Concentration (MIC) • The lowest concentration of antimicrobial agent thatinhibitsbacterial growth/ multiplication • Minimum Bactericidal Concentration (MBC) or Minimum Lethal Concentration (MLC) • The lowest concentration of antimicrobial agent that allows less than 0.1% of the original inoculum to survive
• • • • • • • • • • • • • Antimicrobial susceptibilitytesting using micro-broth dilutions ug/ml 64 32 16 8 4 2 96 well microtiter plate
Broth Dilution Method • Procedure Making dilutions (2-fold) of antibiotic in broth Mueller-Hinton, Tryptic Soy Broth • Inoculation of bacterial inoculum, incubation, overnight • Controls: no inoculum, no antibiotic • Turbidity visualization MIC • Sub culturing of non-turbid tubes, overnight • Growth (bacterial count) MBC
Creating Dilutions Dr.T.V.Rao MD
128 64 32 16 8 4 2 C1 C2 64 32 16 8 4 2 1 C1 C2 Broth Dilution Method Day 1 Add 1 ml of test bacteria (1*106 CFU/ml) to tubes containing 1 ml broth and concentration of antibiotic (mg/l) Controls: C1 = No antibiotic, check viability on agar plates immediately C2 = No test bacteria Bacterial conc.= 5*105 CFU/ml Incubate 35 oC, o/n
64 32 16 8 4 2 1 C1 C2 64 32 16 Broth Dilution Method Day 2 Record visual turbidity Subculture non-turbid tubes to agar plates (use 0.01 ml standard loop) MIC = 16 mg/l 0.01 ml (spread plate), Incubate 35 oC, o/n Day 3 Determine CFU on plates: At 16 mg/ = 700 CFU/ml > 0.1% of 5*105 CFU/ml MBC = 32 mg/l
Broth Dilution Method • 100% of original bacterial conc. • = 5*105 CFU/ml • 0.1% • = [(5*105)*0.1]/100 CFU/ml • = 500 CFU/ml • The bacteria count should be less than 5 CFU on agar plate subcultured with 0.01 ml • 500*0.01 = 5 CFU
Broth Dilution Method are Technically Difficult • Disadvantages : • Only one antibiotic & one organism can be tested each time • Time-consuming • Solutions?? • Agar dilution method • Disc diffusion method • Micro broth dilution method Dr.T.V.Rao MD
Micro broth Dilution Method • Micro dilution plates: • “Micro dilution/ Micro broth dilutions” • 96 wells/ plate: simultaneously performed with many tests organisms/ specimens, less reagent required • Manually prepared • Commercially prepared • Frozen or Dried/ lyophilized • Consistent performance but high cost • May suffer from degradation of antibiotic during shipping and storage
Agar Dilution Method • Procedure • Making dilutions of antimicrobial agent in melted media and pouring plates • One concentration of antibiotic/ plate • Possible for several different strains/plate 64 uGu/ml 32 ug/ml 16 ug/ml
Agar Dilution Method • Procedure • Inoculation of bacterial inoculum (McFarland No. 0.5) • Using a replicating inoculator device called “A Steers-Foltz replicator” • Delivers 0.001 ml of bacterial inoculum • Incubation • Spot of growth MIC 32 ug/ml
Minimal inhibitory concentration • The lowest concentration of antimicrobial agent that inhibits the growth of a bacterium • Interpret: • Susceptible • Intermediate • Resistant Dr.T.V.Rao MD
Clinical Conditions when MICs are Useful • Endocarditis • Meningitis • Septicemia • Osteomyelitis • Immunosuppressed patients (HIV, cancer, etc.) • Prosthetic devices • Patients not responding despite “S” Reports Dr.T.V.Rao MD
Inoculum PreparationMIC Testing (NCCLS Reference Method) • Standardize inoculum suspension • Final inoculum concentration 3 – 5 x 105 CFU/ml (3 – 5 x 104 CFU/well) Dr.T.V.Rao MD
Select Micro titration plate and prepare optimal inoculum Prepare inoculum suspension Micro dilution MIC tray Dr.T.V.Rao MD
Dilute & mix inoculumsuspension Dr.T.V.Rao MD