Genetics, Epigenetics and Dynamic Molecular Memory
Genetics, Epigenetics and Dynamic Molecular Memory. The Basics: Genetics – The G,A,T,Cs of inheritance. Mutations or changes in genotype may result in changes in phenotype. Epigenetics – Heritable chromatin remodeling.

Genetics, Epigenetics and Dynamic Molecular Memory
E N D
Presentation Transcript
Genetics, Epigenetics and Dynamic Molecular Memory
The Basics: Genetics – The G,A,T,Cs of inheritance. Mutations or changes in genotype may result in changes in phenotype. Epigenetics – Heritable chromatin remodeling. Covalent modification of DNA or chromatin proteins can be passed on. Dynamic molecular memory – Stable switching of gene expression; which mimics a mutation in an individual. Relatively stable covalent modification of DNA or chromatin proteins.
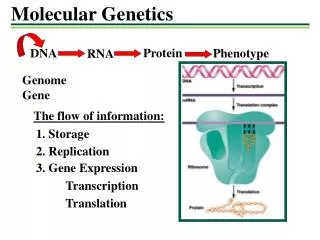
GENETICS Study of one generation to the next. Concerns the chemical nature of genes. Central Dogma: DNA->RNA->Protein • Mutations can change: • DNA -> protein seq. • DNA -> protein folding • Reg. DNA -> expression • Genetic convention: • e.g. CAT -> CGT • a4235g – DNA change • H202R – coding change Changes in the genetic makeup of a population over time and across geographical space.
Meiosis: Simple, but often confusing…
Genetic Animal Models: Outbred = animals with diverse genetic make-up, containing multiple alleles [various lab rats] Inbred Strain = all animals equal to identical twins (isogenic) [C57BL/6, DBA, most lab mice] - homozygous at ALL alleles Recombinant Inbred Strain = inbred offspring from crosses of two inbred strains [BxD] - genetic mix of two strains, but completely homozygous at ALL alleles Congenic strain = region on one chromosome introgressed onto different background strain Consomic strain = one whole chromosome is introgressed from one strain to another Knock out, Knock in = a single gene is removed, completely added or simply changed - however, keep in mind that since two strains are used, linked genes fix Transgenic strain = a gene is added in “trans”; i.e. it can insert anywhere in the genome Line = Selectively bred line of animals…similar to what we consider a “breed”
Picture worth a thousand words: [Consomic strains] [Set of genome-wide congenic strains]
Gene Expression and Chromatin: Constitutive Heterochromatin: chromatin that usually remains condensed in all cells Facultative Heterochromatin: regions that are condensed in some cells, and not in others Euchromatin: open, or less condensed DNA regions X-inactivation results in Barr bodies with different X chromosomes shut off in each cell. Since this is done early in development, a patch effect is seen.
Histone modifications alter local chromatin structure and changes access of transcriptional machinery to bind DNA: Euchromatin is not simply constitutively active: Nucleosome = Octet of 2x (H2A, H2B, H3 and H4) • Silent Chromatin: • - Hypoacetylation of • histone H3 and H4 • amino-terminal tails • - DNAse resistant • Methylated DNA • serves as a recruiting • point for deacetylation • enzymes • - H3 can be dynamically • replaced by varient • H3.3, an active histone • protein Chromosomal regions such as teleomeres that are tethered to the periphery are silenced…but recent studies suggest that localization specific silencing is more complex.
Eukaryotic regulation of gene expression: Analysis of Bergeson lab microarray data suggests regional cis-control:
Epigenetics: A change in phenotype that is heritable but does not involve DNA mutation. (Gottschling. Epigenetics, 2007) • 1. Position effect variegation (Muller, 1941; McClintock, 1951 on transposition) • 2. Trans-control of expression (for example λbacteriophage switching, Ptashne, 2004) • 3. X – Inactivation, Genomic Imprinting & Facultative Heterochromatin • 4. Prion disease • 5. Cytoplasmic Inheritance / Genetic Maternal Effect • microRNA regulation of mRNA content and translation of proteins • ________________________________________________________ • Dynamic Nuclear Organization • Dynamic DNA modification and the “Histone Code”
Position Effect Variegation: The Avy allele: map of the A locus and range of phenotypes in isogenic Avy mice:a, Avy has an IAP (subtype II, striped box) inserted in the pseudoexon 1A of the locus, with the direction of transcription from the LTRs (arrowhead) opposite to that of the A promoters. Hair-cycle−specific non-coding exons (open boxes), coding exons (filled boxes) and an interrupted inverted repeat (grey bar arrow) are indicated. The locus is not shown to scale (100 kb separates the insertion site and hair-cycle−specific promoters). Transcription originating in a cryptic promoter (arrowhead) in the 3' LTR of the IAP in the Avy allele results in constitutive expression of agouti in multiple tissues1, 2, 4, 25. b, Isogenic C57BL/6 Avy/a mice show a continuum of phenotypes ranging from completely yellow, through degrees of yellow/agouti mottling, to completely agouti (termed pseudoagouti because the mice are isogenic with fully yellow mice and not genetically agouti). The extent of the yellow coat colour correlates closely with adult body weight. Yellow mice have pancellular agouti expression driven by the inserted IAP. Mottled mice are mosaics of cells that have or lack ectopic expression driven by the IAP. Pseudoagouti mice lack expression from the cryptic promoter, so that A is regulated by its hair-cycle promoters, and these mice have the wild-type coat colour and normal body weight1, 2, 3. Morgan et al., Nature Genetics, 1999.
Dynamic Nuclear Organization: Chromatin Mobility within the Nuclear Architecture affects Gene Expression Organization of the nucleus:
Dynamic Molecular Memory • Covalent DNA modification • The “Histone Code”
DNA Methylation: at CpGdinucleotides • Used during replication to tag the old strand so repair enzymes can fix mutations • 2. Mechanism of X-inactivation (controlled by XIST gene expression, Willard et al., 1993) • Although always thought be a permanent covalent change, new evidence shows that demethylation does, in fact, take place making changes plastic. • Hypomethylation – active • Hypermethylation – silent
DNA Methyltransferases: DNMT1 – Maintenance of methylation DNMT2 – has weak activity with unknown function DNMT3A – de novo methylation DNMT3B – de novo methylation DNA Demethylation: Understood to be “active” removal, but mechanism remains to be elucidated. (Histone demethylases have been identified.)
Covalent Modification of DNA Regulates Memory Formation: Miller and Sweatt, Neuron, 2007 Fear conditioning increases DNA methylation enzymes. Proposed molecular mechanism of long-term memory
Genotype Specific Change in Drinking Associated with Decrease in Dnmt3b: Bergeson and Blednov: preliminary data
E-phys data from Harris Lab. Evidence Age-Specific Epigenetic Regulation by Binge Alcohol Drinking: