Comprehensive Analysis of CH4, CH2, and NO3 Functional Groups in Java Style Representation
This document explores the molecular structures of various chemical species including CH4, CH2, and NO3 through Java style representation. It emphasizes their geometrical and electronic configurations, along with lone pair and charge distributions. The analysis is supported by methods that detail electron styles, bonding characteristics, and the interaction of hydrogen and nitrogen atoms within the compounds. This serves as a vital resource for understanding molecular chemistry in computational chemistry frameworks.

Comprehensive Analysis of CH4, CH2, and NO3 Functional Groups in Java Style Representation
E N D
Presentation Transcript
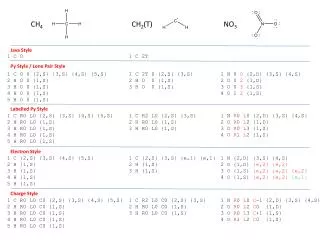
CH4 CH2(T) NO3 Java Style 1 C 0 1 C 2T Py Style / Lone Pair Style 1 C 0 0 {2,S} {3,S} {4,S} {5,S} 2 H 0 0 {1,S} 3 H 0 0 {1,S} 4 H 0 0 {1,S} 5 H 0 0 {1,S} 1 C 2T 0 {2,S} {3,S} 2 H 0 0 {1,S} 3 H 0 0 {1,S} 1 N 0 0 {2,D} {3,S} {4,S} 2 O 0 2 {1,D} 3 O 0 3 {1,S} 4 O 1 2 {1,S} Labelled Py Style 1 C R0 L0 {2,S} {3,S} {4,S} {5,S} 2 H R0 L0 {1,S} 3 H R0 L0 {1,S} 4 H R0 L0 {1,S} 5 H R0 L0 {1,S} 1 C R2 L0 {2,S} {3,S} 2 H R0 L0 {1,S} 3 H R0 L0 {1,S} 1 N R0 L0 {2,D} {3,S} {4,S} 2 O R0 L2 {1,D} 3 O R0 L3 {1,S} 4 O R1 L2 {1,S} Electron Style 1 C {2,S} {3,S} {4,S} {5,S} 2 H {1,S} 3 H {1,S} 4 H {1,S} 5 H {1,S} 1 C {2,S} {3,S} {e,1} {e,1} 2 H {1,S} 3 H {1,S} 1 N {2,D} {3,S} {4,S} 2 O {1,D} {e,2} {e,2} 3 O {1,S} {e,2} {e,2} {e,2} 4 O {1,S} {e,2} {e,2} {e,1} Charge Style 1 C R0 L0 C0 {2,S} {3,S} {4,S} {5,S} 2 H R0 L0 C0 {1,S} 3 H R0 L0 C0 {1,S} 4 H R0 L0 C0 {1,S} 5 H R0 L0 C0 {1,S} 1 C R2 L0 C0 {2,S} {3,S} 2 H R0 L0 C0 {1,S} 3 H R0 L0 C0 {1,S} 1 N R0 L0 C-1 {2,D} {3,S} {4,S} 2 O R0 L2 C0 {1,D} 3 O R0 L3 C+1 {1,S} 4 O R1 L2 C0 {1,S}