Smooth muscle activation and regulation
300 likes | 1.07k Vues
Smooth muscle activation and regulation. Actin polymerization and depolymerization. Organization of smooth muscle cells. Contractile apparatus : actin and myosin filaments The ratio of thin to thick filaments is much higher in smooth muscle (~15:1) than in skeletal muscle (~6:1).

Smooth muscle activation and regulation
E N D
Presentation Transcript
Smooth muscle activation and regulation Actin polymerization and depolymerization
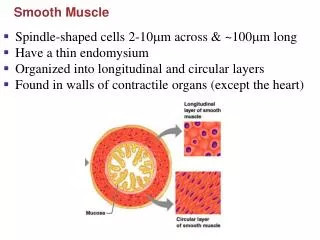
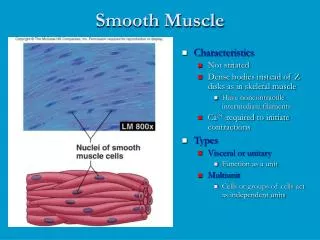
Organization of smooth muscle cells • Contractile apparatus : actin and myosin filaments • The ratio of thin to thick filaments is much higher in smooth muscle (~15:1) than in skeletal muscle (~6:1). • Cytoskeleton compartment : actin filaments and intermediate filaments • Desmin and vimentin : two major intermediate filament proteins • The sarcomeric structure is poorly understood due to the lack of visible cross striations within the cells in the light or electron microscope.
The model of malleable sarcomeric structure of contractile filament
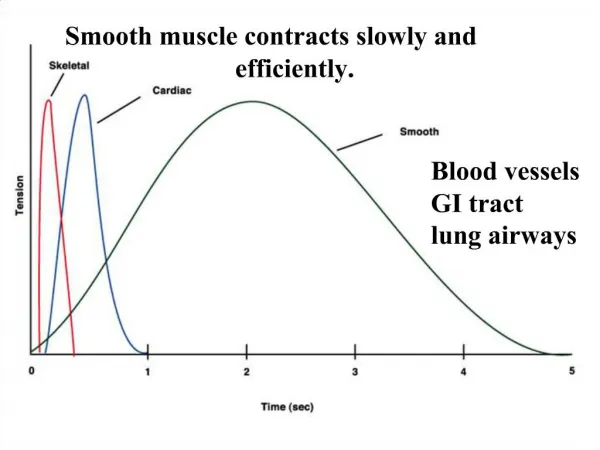
Smooth muscle contraction • Sharing the mechanism of the sliding-filament, cross-bridge cycling as proposed in striated muscle. • Optimize the contractile filament overlap at any cell length within its physiological range and to maintain the ability to generate maximal force over that length range. • Contractile activation involves cytoskeleton rearrangements • Dynamic remodeling of the actin filament lattice within cellular microdomains in response to local mechanical and pharmacological events enables the cell to maintain its external environment. • As the contraction occurs, the cytoskeletal lattice stabilizes, solidifies, and forms a rigid structure for transmission of tension generated by the interaction of myosin and actin. The integrated molecular transitions take place within the contractile cycle.
Thick filament regulation of smooth muscle contraction • The Ca2+-dependent phosphorylation/dephosphorylation of MRLC by myosin light chain kinase (MLCK)/ myosin light chain phosphatase (MLCP) is thought to control the contraction-relaxation cycle of smooth muscle. • Smooth muscle myosin similar to striated muscle counterpart contains two heavy chains (MHC) and two pairs of light chains, one 20-kD regulatory light chains (MRLC) and the other 17-kD essential light chains (MELC). • Activated by Ca2+-calmodulin, MLCK causes phosphorylation of serine-19 and/or threonine-18 at MRLC, and MRLC phosphorylation increases myosin’s actin-activated ATPase activity at least 100-fold • Once phosphorylated, the myosin cross-bridge can bind to actin, generating force by cross-bridge cycling. • On the other hand, dephosphorylation is brought about by MLCP.
Thin filament regulation of smooth muscle contraction • The demonstration of Ca2+ sensitivity of myosin ATPase activity in preparations containing skeletal muscle myosin and smooth muscle thin filament • Reports that cross-bridge cycling rates can vary without detectable changes in MRLC phosphorylation • Reports of dissociations between MRLC phosphorylation and tension • Under some conditions, unphosphorylated cross-bridges are not completely turned off, which indicated that force is also regulated by thin filament associated proteins
Actins: • At least six tissue-specific isoforms: a-skeletal, a-cardiac, and a-vascular; g-enteric and g-cytoplasmic; and b-cytoplasmic actins • Highly homologous in their primary structure with most sequence differences in the N-terminal region of the molecule. This part of the molecule is not functionally involved in filament assembly but is the site for interaction with various actin binding proteins • The a-, b- and g- isoforms can be resolved by isoelectric focusing, proceeding from the most acidic to the least acidic • The smooth muscle actins have been associated with the contractile filaments, whereas the cytoplasmic actins have been associated with the non- contractile cytoskeleton and sub- plasmalemma cortex.
Tropomyosin (TM) • From four highly conserved genes a-, b-, g-, d-TM , via alternative splicing, give rise to more than 40 isoforms • a-TM is a major isoform in smooth muscle with minor b-TM to form a homo- or hetero-dimer that binds along the major groove of actin filament in a head-to-tail manner • In vitro studies have implicated TMs in the stabilization of the actin cytoskeleton by protecting actin filaments from the severing action of gelsolin and the depolymerizing action of ADF/cofilin • X-ray studies have suggested that the activation of smooth muscle leads to the movement of TM in a manner similar to that in striated muscle • The movement of smooth muscle TM by myosin binding is more easily facilitated by phosphorylated myosin than unphosphorylated myosin, providing for possible cross talk between thick and thin filament regulation. • TM is necessary for full inhibition of actomyosin ATPase activity by caldesmon
Caldesmon (CaD): • CaD is an actin, Tm, myosin, and calmodulin (CaM) binding protein. • Two isoforms are produced by a single gene through alternative splicing to generate a smooth muscle form, h-CaD with high molecular weight of 130~140 kD and a non-muscle l-CaD with low molecular weight of 60~90 kD • The difference between h- and l-CaD is a highly charged repeating sequence, corresponding to a 35 nm-long single helical region that separates the N-terminal domain from the C-terminal domain of h-CaD.
CaD • An elongated protein containing two relatively compact domains at the N- and the C-terminal ends • The C-terminal domains are responsible for actin binding and inhibition of myosin ATPase activity in vitro. • Binding of CaM or phosphorylation of sites between the two C-terminal actin binding domains can reverse some of the inhibitory actions of CaD in vitro. • The N-terminal half of the molecule has been shown to bind myosin and, in vitro, tether myosin to actin in conjunction with C-terminal actin binding domains of CaD
Calponin (CaP): • CaP is an actin binding protein that also binds to CaM, myosin, desmin, and phospholipids • There are three different isoforms encoded by separate genes and expressed in smooth muscle (h1 basic CaP), cardiac muscle (h2 neutral CaP), and non-muscle cells (acidic CaP), respectively. • In smooth muscle, CaP interacts with F-actin and inhibits the actomyosin Mg-ATPase activity in vitro. When phosphorylated in vitro by either CaMKII or PKC, the inhibitory action of CaP on the actin-myosin interaction is reversed • Smooth muscle CaP is an elongated molecule (~MW: 32 kD) of 18 nm long • The domain structure containing a CaP homology domain (CH), followed by a TnI-like domain, and three C-terminal repeat sequence. The CH domain in smooth muscle CaP is thought to localize the signaling molecules to the actin cytoskeleton since this domain can bind to ERK. • Both TnI-like domain and C-terminal repeat can bind to actin.
Actin polymerization and depolymerization • Actin exists in two forms, globular (G) monomer and filamentous (F) polymer. • G-actin contains bound ATP, which, on polymerization, is transformed into F-actin-bound-ADP and Pi • At intermediate concentration of free G-actin subunits, the filaments loose actin-ADP at the minus–end (pointed end) and assemble actin-ATP at the plus-end (barbed end). This phenomenon is called “treadmilling” of actin filament polymerization/depolymerization. • The treadmilling of actin filament is the basic mechanism for motility in nonmuscle cells
Actin associated proteins in actin filament polymerization and deplymerization • Proteins that bind to G-actin: -profilin, thymosin • Proteins that nucleate F-actin : -formin, Arp2/3 (also branching effect) • Proteins that bind to F-actin ends and control polymerization: - cofilin, gelsolin : severing, and capping at the plus end - gCAP39, capZ : capping at the plus end - tropomodulin : capping at the minus end - severin : severing, and capping - villin : crosslinking, severing, and capping • Proteins that attach actin to the plasma membrane: Ezrin/Radixin/Moesin (ERM) protein family • Proteins that crosslink/branch F-actin: filamin, fimbrin, a-actinin, fascin, spectrin
Arp3 47 Arp2 42 21 20 34 40 16 Non-zero crosslink Two hybrid Zero-length crosslink (Higgs and Pollard, 1999) Arp2/3 complex: downstream target of multiple signaling pathways leading to actin assembly • Seven conserved subunits including Arp2 and Arp3 (Machesky et al., 1994). • Generates new actin filaments in a stimulus-responsive and spatially controlled fashion.
Physiological functions • Using the methods of DNase inhibition and actin affecting drugs (phalloidin, cytochalasin, and latrunculin), Mehta and Gunst first found that the G-actin content was 30 % lower in extracts of muscle strips activated with acetylcholine than in extracts from unstimulated muscle strips. The decrease in G-actin in response to contractile stimulation was prevented by inhibitors of actin polymerization with no effect on MRLC phosphorylation. • Actin polymerization in smooth muscle is functionally related to contractile activity under physiological conditions • Bárány and his colleagues found a rapid exchange of the G-actin bound-ATP in intact arterial smooth muscle, indicating that dynamic rearrangement of actin filament occurs in intact smooth muscle. • All these studies have suggested that polymerization of G-actin monomer into filamentous F-actin may play an important role in contractile activation in smooth muscle independent of activation of myosin ATPase activity in response to MRLC phosphorylation.
Contractile stimulation inducing the formation of linkages betweenthe cytoskeleton and integrin proteins • Receptor coupled G-protein (Rho) activation by extracellular stimulation of carbachol may involve in actin reorganization in human airway smooth muscle. • Signaling cascade of integrin activation to remodeling of cytoskeleton in regulation of smooth muscle contraction • Cholinergic receptor stimulation recruits actin- and integrin- binding proteins (e.g. a-actinin, vinculin, talin, focal adhesion kinase, and paxillin) from the cytoplasm to the membrane cytoskeleton. • Depletion of paxillin and focal adhesion kinase led to loss of force in tracheal smooth muscle tissues
Linkages betweenthe cytoskeleton and integrin proteins • Nonphosphorylatable paxillin mutants in tracheal muscle inhibits tension development and actin polymerization, without affecting MRLC phosphorylation • Down-regulation of profilin, an actin regulatory protein, was found to attenuate the force development of arterial smooth muscle as well • Membrane cytoskeletal linker proteins (e.g. a-actinin, vinculin, talin etc.) and integrin associated proteins (e.g. FAK, and paxillin) plays an important role in reorganization of actin filament polymerization and depolymerization in the membrane/cytoskeletal compartment during smooth muscle contraction. • Contractile stimulation could induce formation of the structural linkage between the cytoskeleton and integrin proteins that mediate tension transmission between the contractile apparatus and the extracellular matrix in smooth muscle.
Actin binding protein regulation of actin filament • Formin and Arp 2/3 complexes (actin related proteins 2 and 3, seven strongly associated subunits) are the two nucleating proteins that facilitate the polymerization of actin filament. • While the formin protein induces a linear form of actin filament, the Arp 2/3 protein complex branches the actin filaments to form a network within the cells. • Profilin is an actin binding protein to promote the nucleotide exchange of G-actin and to increase the polymerization rate of actin filament formation. • Cofilin associated with actin depolymerization factor (ADF/cofilin), gelsolin, severin, are known to cut off the actin filament and involved in depolymerization of actin filament.
Wiskott-Aldrich syndrome protein (WASp) • Originally described in fibroblasts could induce actin polymerization in connection with the small GTPase cdc42 • WASp family proteins bind to Arp2/3 complex and induce Arp2/3 complex activation and the nucleation of actin filaments • WASp contains a family of homologous proteins found in different cells. • All families of WASp contain a conserved region at the C-terminus with a proline-rich (PRO) region followed by one or two WH2 (also called V for verproline homology) domain, a central (C) region and an acidic (A) domain. The PRO region binds profilin and SH3-containing adaptor proteins, WH2 domain for binding G-actin, and A domain for Arp 2/3 binding. On the other hand, two domains at the the N-terminus, the GBD domain binds small G-protein Cdc42, and WH1 for F-actin binding.
B A WH1 Pr o Bee 1 V C B Pr o A WH1 Scar/WAVE V C WASP B GBD A Pr o WH1 V C N-WASP GBD A WH1 B Pr o VV C PIP2 F-Actin G-Actin Cdc42 SH3 Arp 2/3 (Grb2, Nck, IcsA Fgr, Fyn) Domain structures of WASp family protein
Mechanism • In the unstimulated conditions, the WASp molecule has the inhibitory interaction between the N- and C-terminus and burries the binding site for Arp 2/3. Upon stimulation, Cdc42 binding to GBD domain activates WASp, and unfold the N-and C-terminal domains of the molecule, and expose the binding site for Arp 2/3, finally initiate the actin filament polymerization .
Signaling pathway by WASp regulation in smooth muscle • The signaling cascades by profilin and its upstream regulator, p130 Crk-associated substrate are necessary for regulation of actin filament polymerization and force generation in arterial smooth muscle in response to contractile stimulation. • Small GTPase Cdc42 regulates actin polymerization and also tension development during contractile stimulation of tracheal smooth muscle. • Crk II regulation of N-WASp-mediated activation of the Arp2/3 complex is also necessary for actin polymerization and tension development in response to muscarinic stimulation in tracheal smooth muscle. • These studies provide evidence showing that the signaling pathway by the regulation of WASp activation upon contractile stimulation cause the reorganization of the actin filament network and smooth muscle contraction. • Newly formed actin filaments may localize beneath the cell membrane (dense bodies) and may be critical for the transmission of tension from the contractile apparatus to adhesion sites on the membrane during contractile stimulation.