HERBAL IN PHARMACEUTICAL INDUSTRY
780 likes | 2.59k Vues
HERBAL IN PHARMACEUTICAL INDUSTRY. PRESENTED BY DILLIP KUMAR JENA. Dept .of pharmaceutics and pharmaceutical Technology L.M.College of pharmacy. INRODUCTION Approaches to Herbal Formulation Development Standardization of Herbs Marketing of Herbal Formulations Guidelines for Using Herbs

HERBAL IN PHARMACEUTICAL INDUSTRY
E N D
Presentation Transcript
HERBAL IN PHARMACEUTICAL INDUSTRY PRESENTED BY DILLIP KUMAR JENA Dept .of pharmaceutics and pharmaceutical Technology L.M.College of pharmacy
INRODUCTION • Approaches to Herbal Formulation Development • Standardization of Herbs • Marketing of Herbal Formulations • Guidelines for Using Herbs • List of References • Questions
Results of Patient Survey Questionnaire 3106 patients were surveyed…….. • 22% of presurgical patients reported the use of herbal remedies • 51% used vitamins • Women and patients 40-60 more likely to use herbal medicines
Why Herbal Products In Demand • Placebo effect - Mind over Matter • Placebos do what you think they will do • Placebos work about one-third of the time
Two Sides to Herbs • Assumed herbs have only benefits • Two sides to herbs • Most herbs have mostly placebo effects with minimal good and bad effects • Some herbs are more beneficial than harmful, while others are more harmful than beneficial
Herbs Are Safe • Herbs are the source of some potent pharmaceutical medications • extremely useful, and potentially fatal • herbal products are safe because they are natural • can be harmful if consumed improperly or in excessive amounts
Not Well-Tested • Because herbs are classified as a “dietary supplements” not “food” or “drugs” they do not have to have go through the pre-market testing that drugs and food additives do
Lack Quality Control • Because herbs are classified as “dietary supplements” they do not have the quality control standards “drugs” do • There have been problems with • Identification • Quality • Contamination
Potency Imprecise • Potency of an herbal products can vary • Plant variety • Part of plant used • Environmental conditions grown • How processed • Some standardized herbal products with known concentrations of active ingredients
Interact With Medications • Herbs contain active ingredients that act like drugs • Interact with other herbs or pharmaceutical medications
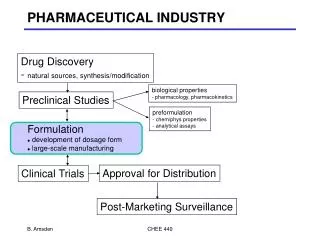
Approches to Herbal Formulation Development As a formulator one has two major options • The conventional or traditional dosage form, • Modern concept
Conventional or traditional dosage form • Ark - Obtained by distillation of water soaked drugs in ark-yantra • Asva/Arista - Drug is soaked in solution of jaggery and allowed to ferment. • Avaleha/Pak - Decoctions(kasayas) or infusions (kalkas) of drug is repeatedly boiled and condensed with jaggery • Bhasma - Prepared by heating (oxidation) of drug
Conventional or traditional dosage form… • Churna - A form of powder • Ghrita - Ghee is boiled with decoctions(kasayas) or infusions (kalkas) of drug. • Gutika/Bati/Pill - Pulverized drug admixed with honey and made up into Gutika or Bati or Pill. • Kwatha - Coarse powder of drugs or in combination for kasayas.
Conventional or traditional dosage form… • Lepa - Paste for external applications • Satva/Dravya - Drug is cut into small pieces and macerated in water overnight, strained through cloth. process is repeated and the supernatant liquid (Satva) is dried and powdered. • Taila - Taila is boiled with decoctions (kasayas) or infusions (kalkas) of drug.
Sweet Proteins • Low calorie high intensity natural sweeteners • Sweet protein plants native to West Africa • Thaumatin (T. danielli) – 3000 X sweeter than sugar • Brazzein (P. brazzeana) - heat stable protein; j’oublie • Miraculin (R. dulcifica) - alter sour taste to sweet
Modern concept of dosage form • Tablet (Plain or sugar coated or film coated) • Lozenges, • Capsule (hard or soft), • Liquid orals (suspension or emulsion or syrup), • External application (cream or ointment) or • Sterile preparations (injectables or eye or ear drops), etc.
Choice of Herbal Ingredients After finalizing the dosage form one intend to prepare, the herb or herb part is selected • Based on traditional literature available • By phytochemical investigations of the herb or herb part • Clinical trials/bioactivity studies of herb or herb part
Choice of Herbal Ingredients • Powders of the herb or herb part so selected may be used as such, if they are potent enough. • Usually, processing of herb or herb part is desired to obtain a herbal drug of suitable/sufficient potency. like • Extraction, • Distillation, • Fermentation, etc. :
Factors affecting the choice of processing method • Shodhan • Proper choice of solvent • Choice of process • How to admix Herb Extract into the Formulation • Number of HerbalIngredients
Shodhan - which involves the detoxifying/purifying the herb or herb part with cow urine, to make the herb safe. • Proper choice of solvent - keeping in view the solubility, cost, toxicity, taxes and Law (Drug Law and others).
Choice of process - manufacturing process/equipments should keep in view the thermostability of the active constituent(s) present in the herb or herb part since the activity of the herb is generally attributed to the complete extract of the herb or herb part.
Number of Herbal Ingredients : • This must be kept to the minimum subject to the therapeutic efficacy in minimum dosage, toxicity and safety of the formulation.
Herbal Tablet • The dried herb extract is admixed with starch, tragacanth, gums, to act as binders/ disintegrators and lubricants, as in the case of allopathic formulations. • The tablet may then be sugar coated or film coated or marketed plain, as such.
Herbal liquid orals • Herb or herb part, coarsely or finely powdered is extracted with water (by hot or cold process), consistent agitation and filtration. • Extract has to be treated carefully with respect to its pH, which must allow chemical and physical stability of the product uniformly. .
Procurement of proper herb • Names of the herbs mentioned in the scriptures and ancient literature does not match the local or vernacular names at places, which at times does not allow proper identification. • The nomenclature may change from region to region of the same drug. • Example ‘Haritaki’ (fruit of Terminalia chebula) of which seven varieties originating from different parts of the country, are attributed with different therapeutic properties.
Identification of the herb • Macroscopy • Microscopy
Identification of the herb • Fluorescence studies : • The fluorescence is unique under UV light for certain herbs. • Qualitative test using UV, TLC, HPTLC :
Checking the quality of the herb The herb is subjected to the following various parameters singly or in combination to ensure the quality of the herb • Extractive values • Alcohol and water soluble extractives do give an idea of the quality of the herb. • Ash value • The Total ash, acid insoluble ash and water soluble ash are helpful tools in ensuring quality herb
Checking the quality of the herb • Foreign matter • Any non-specified part of the plant or matter should not exceed the prescribed limit set to ensure quality herb. • Chemical Assay • Specific assays for active principles viz. total alkaloids, glycosides, resins, tannins, saponins, volatile oil contents, etc. are carried out by chemical means. .
Checking the quality of the herb • Instrumental Analysis : • Microbial Contamination
Toxicity studies • The herbs mentioned in the scriptures and ancient literature should be first tested for toxicity, if any, and not just be taken, since scientific data may not be available for them
Points to ensure quality of herbs • Herbs procured are collected only when their active principles are maximum • Unwanted foreign material free herb is made available • Herbs procured should be graded according to quality • Drying of herb should be under controlled conditions, with moisture usually below 9-10% (excess would encourage fungal growth)
Parameters of the Finished Product • The finished product should be standardized in terms of • shelf life, • colour, • taste, • toxicity studies and • biological assay
WHO Guidelines – Assessment for modern herbal medicines • Pharmaceutical assessment • Crude plant material • Plant preparations • Finished product • Stability • Activity • Evidence required to support indication • Combination products
PACKAGING AND STORAGE • It provides an image of the final product and serves as an identity of certain medicines. • Packaging and storage is dictated by GPP guidelines for modern medicines. • Material used for the packaging of medicines differ for both disciplines. • Storage practices for both discipline differ, with modern medicine being more controlled with the methods used.
MARKETING • The industry is largely informal and undeveloped despite it size, economic value and cultural importance. • There is no law regulating the market due to lack of corporation between traditional healers and health professionals. • Healers market themselves mainly through their patients.
MARKETING • Phytomedicines are marketed, but there is strict control exercised. The information given must be reliable, accurate, truthful, informative and capable of substantiation. • Promotional material shall not be designed to disguise the general public of the true nature of the medication. • Marketing strategies must contain information such as the active ingredients, indications, dosing, warnings and precautions.
Patients Use Herbs but Doctors are Reluctant to Prescribe Them. Why? • Knowledge deficit • Tradition • Real concerns about product safety, drug-herb interactions • Unnecessarily high concerns about liability
Guidelines For Using Herbs • Purchase standardized herbal products • specified amount of active ingredient • reputable company • provide the herb’s botanical scientific name • provide warnings about potential side effects • provide possible interactions with pharmaceutical medications
Guidelines For Using Herbs • Stop taking an herb if any side effect develops such as allergy, stomach upset, skin rash or headache • Herbal products are not recommended for: • pregnant or lactating women • infants or children under six years of age
Echinacea St. John’s Wort Valerian Ephedra Some Very Popular Herbs Garlic Ginkgo Kava
List of Reference • UTMB— http://atc.utmb.edu/altmed —Resources: HealthNotes Online • NIH--- www.nccam.nih.gov • American Botanical Council — www.herbalgram.org • Herb Research Foundation— www.herbs.org • Duke’s Phytochemical and Ethnobotanical Database http://www.ars-grin.gov/duke