Chordata
taksonomi chordata

Chordata
E N D
Presentation Transcript
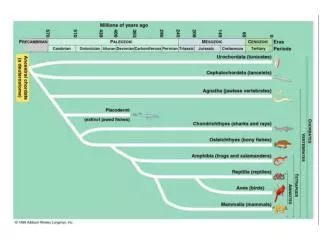
Final PDF to printer 17 Chordata: Urochordata and Cephalochordata These tunicates (Clavelina picta, subphylum Urochordata) look nothing like members of the phylum Chordata, but they are! Careful examination of their larval stage reveals all the features that are hallmarks of the phy- lum. The pair of openings seen on each tunicate are siphons, which are the openings for incurrent and excurrent water that circulates through this filter-feeding animal. Diane R. Nelson Chapter Outline Evolutionary Perspective Phylogenetic Relationships Phylum Chordata Subphylum Urochordata Subphylum Cephalochordata Evolutionary Connections 17.1 17.2 17.1 EVOLUTIONARY PERSPECTIVE 17.3 LEARNING OUTCOME 1. Describe the evolutionary relationships of the Chordata to other animal phyla. Some members of the phylum discussed in this chapter are more familiar to you than members of any other group of animals. This familiarity is not without good reason, for you yourself are a member of this phylum—Chordata. Other members of the Chordata, however, are much less familiar. During a walk along a seashore you could see animals clinging to rocks exposed by low tide. At first glance, you might describe them as jellylike masses with two openings at their unattached end. Some live as solitary individuals; oth- ers live in colonies. If you handle these animals, you may be rewarded with a stream of water squirted from their openings. Casual observations provide little evidence that these small filter feeders, called sea squirts or tunicates, are chordates. However, detailed studies have made that conclusion certain. Tunicates and a small group of fishlike animals called lancelets are often called the invertebrate chordates because they lack a vertebral column (figure 17.1). Phylogenetic Relationships Animals in the phylum Chordata share the classic deuterostome characteristics shown in figures 8.11 and 16.19 with echinoderms and hemichordates as well as pharyngeal slits and a host of molecular characters. There is little doubt that members of the Chordata and other deuterostomes arose from a common ancestor during the early to mid-Cambrian period (see figure 17.1and appendix B). Diverse adult body forms within deuterostomia have made evolu- tionary pathways leading to Chordata difficult to discern. As discussed later in this chapter, evidence from paleontological, developmental, and molecular data is mounting that illumi- nates our understanding of the origin of the chordate lineage. miL2218x_ch17_303-312.indd 303 10/21/22 05:06 PM
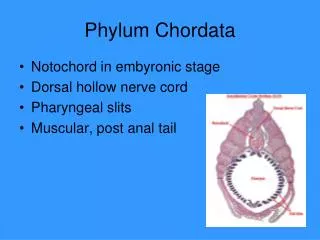
Final PDF to printer 304 CHAPTER SEVENTEEN Animalia Basal Phyla Spiralia Ecdysozoa Deuterostomia Protists Siphons FIGURE 17.1 Evolutionary Relationships of Chordata to Other Animals. This figure shows a widely accepted interpretation of the relationships of the Chordata to other members of the animal kingdom (see appendix A). The relationships depicted here are based on evidence from developmental and molecular biology. Chordates are placed within the Deuterostomia along with the Xenoturbellida, Echinodermata, and Hemichordata. This tunicate, or sea squirt (Polycarpa aurata), is an invertebrate chordate that attaches to substrates in marine environments. Note the two siphons for circulating water through a filter-feeding apparatus. Dr. Dwayne Meadows/NOAA/NMFS/OPR extend posteriorly into a postanal tail, and an endostyle or thyroid gland 3. Complete digestive tract 4. Ventral, contractile blood vessel (heart) 5. Muscles of the body well organized into paired blocks of muscles called myomeres. The combination of five characteristics listed in number 2 is distinctive of chordates, and these characteristics are discussed fur- ther in the paragraphs that follow (figure 17.2). Members of the Chordata possess paired trunk muscles that extend posterior to the anus into a tail. These paired muscles develop in blocks called myomeres (see figure 23.19) that are separated from one another by sheets of connective tissue. These muscles accomplish the side-to-side bending movements of the trunk and tail associated with swimming or other forms of locomotion. (Other nonchordate, tailed animals [e.g., decapod crustaceans] have their anus near the tip of their tail.) The chordate post-anal tail is modified for a variety of functions, including locomotion. It is supported by the notochord (described in the following paragraph) or the vertebral column. The phylum is named after the notochord (Gr. noton, the back + L. chorda, cord), a supportive rod that extends most of the length of the animal dorsal to the body cavity and into the tail. It consists of a connective-tissue sheath that encloses cells, each of which contains a large, fluid-filled vacuole. This arrangement gives the notochord some turgidity, which prevents compression along the anteroposterior axis. At the same time, the notochord is flexible 17.2 PHYLUM CHORDATA LEARNING OUTCOMES 1. Describe the characteristics of members of the phylum Chordata. 2. Compare adult tunicates to the generalized chordate body form. 3. Compare adult cephalochordates to the generalized chordate body form. Although the phylum Chordata (kor-dat′ah) (L. chorda, cord) does not have an inordinately large number of species (about 65,000), its members have been very successful at adapting to aquatic and terrestrial environments throughout the world. Sea squirts, mem- bers of the subphylum Urochordata, are briefly described in the “Evolutionary Perspective” that opens this chapter. Other chor- dates include lancelets (subphylum Cephalochordata) and the vertebrates (subphylum Craniata) (table 17.1). In addition to a host of developmental and molecular characteristics, the major morphological characteristics of the phylum Chordata include the following: 1. Bilaterally symmetrical, deuterostomate animals 2. A unique combination of five characteristics present at some stage in development: notochord, pharyngeal slits or pouches, dorsal tubular nervous system, paired trunk muscles that miL2218x_ch17_303-312.indd 304 10/21/22 05:06 PM
Final PDF to printer Chordata: Urochordata and Cephalochordata 305 TABLE 17.1 CLASSIFICATION OF THE CHORDATA Infraphylum Vertebrata (ver″te-bra′tah) Notochord, nerve cord, postanal tail, and pharyngeal slits present at least in embryonic stages; vertebrae surround nerve cord and serve as primary axial support. Class Petromyzontida (pet′ro-mi-zon″tid-ah) Fishlike; jawless; no paired appendages; cartilaginous skeleton; sucking mouth with teeth and rasping tongue. Lampreys. Class Chondrichthyes (kon-drik′thi-es) Fishlike; jawed; paired appendages and cartilaginous skeleton; no swim bladder. Skates, rays, sharks. Class Actinopterygii (ak″tin-op′te-rig-e-i) Bony fishes having paired fins supported by dermal rays; basal portions of paired fins not especially muscular, tail fin with approximately equal upper and lower lobes (homocercal tail); blind olfactory sacs; pneumatic sacs function as swim bladder. Ray-finned fishes. Class Sarcopterygii (sar-kop-te-rig′e-i) Bony fishes having paired fins with muscular lobes; pneumatic sacs function as lungs; atria and ventricles at least partially divided. Lungfishes, coelacanths, and tetrapods.** (Class) Amphibia (am-fib′e-ah) Skin with mucoid secretions; possess lungs and/ or gills; moist skin serves as respiratory organ; aquatic developmental stages usually followed by metamorphosis to an adult. Frogs, toads, salamanders. (Class) Reptilia (rep-til′e-ah) Dry skin with epidermal scales; amniotic eggs; terrestrial embryonic development. Snakes, lizards, alligators. (Class) Aves (a′vez) Feathers used for flight; efficiently regulate body temperature (endothermic); amniotic eggs. Birds. (Class) Mammalia (mah-ma′le-ah) Bodies at least partially covered by hair; endothermic; young nursed from mammary glands; amniotic eggs. Mammals. Phylum Chordata (kor-dat′ah) Occupy a wide variety of marine, freshwater, and terrestrial habitats. A notochord, pharyngeal slits, a dorsal tubular nerve cord, a postanal tail, and an endostyle or thyroid gland are all present at some time in chordate life histories. About 65,000 species. Subphylum Urochordata (u″ro-kor-da′tah) Notochord, nerve cord, and postanal tail present only in free- swimming larvae; adults sessile, or occasionally planktonic, and enclosed in a tunic that contains some cellulose; marine. Sea squirts or tunicates. Class Ascidiacea (as-id″e-as′e-ah) All sessile as adults; solitary or colonial; colony members interconnected by stolons. Sea squirts (Ascidia, Ciona). (Ascidiacea is comprised of four clades: Stolidobranchia, Molgulidae, Phlebobranchia, and Aplousobranchiata.) Class Appendicularia (a-pen″di-ku-lar′e-ah) (Larvacea) (lar-vas′e-ah) Planktonic; adults retain tail and notochord; lack a cellulose tunic; epithelium secretes a gelatinous covering of the body. Appendicularians (Fritillaria). Class Thaliacea (tal″e-as′e-ah) Planktonic; adults are tailless and barrel shaped; oral and atrial openings are at opposite ends of the tunicate; muscular contractions of the body wall produce water currents. Salps (Salpa, Thetys). Subphylum Cephalochordata (sef″a-lo-kor-dat′ah) Body laterally compressed and transparent; fishlike; all five chordate characteristics persist throughout life. Amphioxus (Branchiostoma). Subphylum Craniata (kra″ne-ah′tah)* Skull surrounds the brain, olfactory organs, eyes, and inner ear. Unique embryonic tissue, neural crest, contributes to a variety of adult structures including sensory nerve cells, and some skeletal and other connective tissue structures. Infraphylum Hyperotreti (hi″per-ot′re′te) Fishlike; skull consisting of cartilaginous bars; jawless; no paired appendages; mouth with four pairs of tentacles; olfactory sacs open to mouth cavity; 5 to 15 pairs of pharyngeal slits; ventrolateral slime glands. Hagfishes. *Members of the Craniata are discussed in chapters 18 through 22. **Cladistic analysis has shown that the following four traditional class designations for tetrapods represent a paraphyletic assemblage. Aves is a lineage within Reptilia, and all tetrapods comprise a clade within Sarcopterygii (see chapters 18 and 19). enough to allow lateral bending when acted upon by the myomeres described above. The elasticity of the notochord provides recoil, which aids in swimming and crawling. In most adult vertebrates, cartilage or bone partly or entirely replaces the notochord. Pharyngeal slits are a series of openings in the pharyngeal region between the digestive tract and the outside of the body. As described previously, pharyngeal slits are an important ancestral character in Deuterostomia, but they are adapted for use in distinc- tive ways by members of this phylum. In some chordates, diverticula from the gut in the pharyngeal region never break through to form an open passageway to the outside. These diverticula are then called pharyngeal pouches. As did other deuterostomes, early chordates used the slits for filter feeding; some living chordates still use them for feeding. Other chordates have developed gills in the pharyngeal pouches for gas exchange. The pharyngeal slits of terrestrial verte- brates are mainly embryonic features and may be incomplete. The tubular nerve cord and its associated structures are one reason for chordate success. The nerve cord runs along the miL2218x_ch17_303-312.indd 305 10/21/22 05:06 PM
Final PDF to printer 306 CHAPTER SEVENTEEN Anterior Water in Nerve ganglion Oral siphon Water out Neural tube Endostyle Atrial siphon Atrium Notochord Dorsal Anus Ventral (b) Stigma Cut edge of body wall Gut Intestine Gonads Heart Stomach Tunic Postanal tail (a) Posterior Pharyngeal slits or pouches Endostyle or thyroid gland (a) Tunic FIGURE 17.2 Chordate Body Plan. The development of all chordates involves the formation of a neural tube, the notochord, pharyngeal slits or pouches, a postanal tail, and an endostyle or thyroid gland. Derivatives of all three primary germ layers are present. (a) Lateral view. (b) Cross section. Atrium Pharynx Endostyle Ventral blood sinus Atrial opening Mucous sheet longitudinal axis of the body, just dorsal to the notochord, and usu- ally expands anteriorly as a brain. This central nervous system is associated with the development of complex systems for sensory perception, integration, and motor responses. The fifth characteristic unique to chordates is the presence of an endostyle or thyroid gland. An endostyle is present on the ventral aspect of the pharynx in tunicates, cephalochordates, and larval lampreys. It secretes mucus that helps trap food particles dur- ing filter feeding. In addition to secreting mucus, the endostyle can bind the amino acid tyrosine. During the metamorphosis of larval lampreys into predatory adults, the larval endostyle is transformed into the thyroid gland. Iodine-containing secretions of the thyroid gland are derived from tyrosine, regulate metamorphosis in many members of the subphylum Craniata, and regulate metabolic rates of all craniates (see chapters 18–22). Stigmas (b) FIGURE 17.3 Internal Structure of a Tunicate. (a) Longitudinal section. Black arrows show the path of water. (b) Cross section at the level of the atrial siphon. Small black arrows show movement of food trapped in mucus that the endostyle produces. as the mouth opening. The second siphon, the atrial siphon, is the opening for excurrent water. The body wall of most tunicates (L. tunicatus, to wear a tunic or gown) is a connective-tissue-like covering, called the tunic, that appears gel-like but is often quite leather-like. Secreted by the epider- mis, it is composed of proteins, various salts, and cellulose. Tunicates are the only animals that produce cellulose. Tunicate cellulose is called tunicin and may have evolved in tunicates as a result of hori- zontal gene transfer from bacterial symbionts early in the urochor- date lineage. Because the tunic is laid down by amebocytes and grows along with the tunciate, the tunic is not molted. Interestingly, tunicin is a highly pure form of cellulose whose properties are being inves- tigated for commercial harvesting for tissue engineering, nanofiltra- tion, and other uses. Tunicin may provide a green substitute for these petroleum-based applications. Some mesodermally derived tissues, including blood vessels and blood cells, are incorporated into the tunic. Rootlike extensions of the tunic, called stolons, help anchor a tunicate to the substrate and may connect individuals of a colony. A second class of urochordates is Appendicularia (approxi- mately 70 species, figure 17.4a). They are planktonic in the pelagic zone (near the surface of the open ocean to just above the seafloor). Adults retain the tadpole body form typical of larval urochordates, and Subphylum Urochordata Members of the subphylum Urochordata (u″ro-kor-dah′tah) (Gr. uro, tail + L. chorda, cord) are the tunicates or sea squirts. Phylogenetic analysis has demonstrated the presence of six urochor- date clades that are currently grouped into three classes. Ascidiacea (four of six clades) is the largest class of tunicates (2,300 species, see table 17.1 and chapter opener andfigure 17.1photographs). They are sessile as adults and are either solitary or colonial. In some locali- ties, tunicates occur in large enough numbers to be considered a dominant life-form. Ascidians attach their saclike bodies to rocks, pilings, ship hulls, and other solid substrates. The unattached end of urochordates contains two siphons that permit seawater to circulate through the body. One siphon is the oral siphon, which is the inlet for water circulating through the body and is usually directly oppo- site the attached end of the ascidian (figure 17.3a). It also serves miL2218x_ch17_303-312.indd 306 10/21/22 05:06 PM
Final PDF to printer Chordata: Urochordata and Cephalochordata 307 (a) FIGURE 17.4 Subphylum Urochordata. (a) Members of the class Appendicularia are planktonic and have a tail and notochord that persist into the adult stage. Oiko labradorensis is shown here without its enclosing test. (b) The thaliaceans, or salps, are barrel-shaped, planktonic urochordates. Oral and atrial siphons are at opposite ends of the body, and muscles of the body wall contract to create a form of weak jet propulsion. Cyclosalpa individuals form ringlike colonies or colonies comprised of chains of rings. A single individual is shown here. Note the blue-green bioluminescent organs on the sides of this individual. The central yellow stripe is the intestinal loop. (a) Russ Hopcroft (b) Cultura RM Exclusive/Alexander Semenov/Getty Images (b) The nervous system of tunicates is largely confined to the body wall. It forms a nerve plexus with a single ganglion located on the wall of the pharynx between the oral and atrial openings (seefigure 17.3a). This ganglion is not vital for coordinating bodily functions. Tunicates are sensitive to many kinds of mechanical and chemical stimuli, and receptors for these senses are distributed over the body wall, espe- cially around the siphons. There are no complex sensory organs. The most obvious internal structures of the urochordates are a very large pharynx and a cavity, called the atrium, that surrounds the pharynx laterally and dorsally (figure 17.3b). The pharynx of tunicates originates at the oral siphon and is continuous with the remainder of the digestive tract. The oral margin of the pharynx has tentacles that prevent large objects from entering the pharynx. Numerous pharyngeal slits called stigmas perforate the pharynx. Cilia associated with the stigmas cause water to circulate into the phar- ynx, through the stigmas, and into the surrounding atrium. Water leaves the tunicate through the atrial siphon. The digestive tract (gut) of adult tunicates continues from the pharynx and ends at the anus near the atrial siphon. The endostyle is a ventral ciliated groove that forms a mucous sheet (seefigure 17.3b). Cilia move the mucous sheet dorsally across the pharynx. Food par- ticles, brought into the oral siphon with incurrent water, are trapped they are usually less than 1 cm in length (excluding the tail). Appen- dicularians secrete a test of protein and cellulose that surrounds the animal (like a bubble) and includes a delicate filter-feeding mesh. The mesh is frequently clogged, and the test is discarded and rebuilt several times each day. These tests contribute to “marine snow,” suspended organic particles that are continually raining down through oceanic water columns. Appendicularians are common and abundant, but they are seldom noticed because they are so very small and delicate. The third class of urochordates is Thaliacea (also approxi- mately 70 species, figure 17.4b). Thaliaceans are free swimming, pelagic filter feeders. Some species (e.g., pyrosomes) form biolumi- nescent colonies of hundreds or thousands of individuals that can be several meters in length. Other species are comprised of solitary individuals. Thaliaceans are also common and comprise an impor- tant component of marine food webs. Maintenance Functions Longitudinal and circular muscles below the body-wall epithelium help change the shape of the adult tunicate. They act against the elasticity of the tunic and the hydrostatic skeleton that seawater con- fined to internal chambers creates. miL2218x_ch17_303-312.indd 307 10/21/22 05:06 PM
Final PDF to printer 308 CHAPTER SEVENTEEN in the mucous sheet and passed dorsally. Food is incorporated into a string of mucus that by ciliary action moves into the next region of the gut tract. Digestive enzymes are secreted in the stomach, and most absorption occurs across the walls of the intestine. Excurrent water carries digestive wastes from the anus out of the atrial siphon. In addition to its role in feeding, the pharynx also functions in gas exchange. Gases are exchanged as water circulates through the tunicate. The tunicate heart lies at the base of the pharynx. One ves- sel from the heart runs anteriorly under the endostyle, and another runs posteriorly to the digestive organs and gonads. Blood flow through the heart is not unidirectional. Peristaltic contractions of the heart may propel blood in one direction for a few beats; then the direction is reversed. The significance of this reversal is not completely understood. One intriguing suggestion is that one-way circulation through the heart would mean that tissues near the end of the circulatory path would only contact blood already depleted of oxygen and nutrients. Periodic reversal of flow through the heart might provide a more uniform distribution of oxygen and nutrients to all tissues. Tunicate blood plasma is colorless and contains vari- ous kinds of amoeboid cells. Ammonia diffuses into water that passes through the phar- ynx and is excreted. In addition, amoeboid cells of the circulatory system accumulate uric acid and sequester it in the intestinal loop. Pyloric glands on the outside of the intestine are also thought to have excretory functions. Notochord Nerve cord Incurrent siphon Endostyle Stigma Oral (incurrent) siphon Atrial (excurrent) siphon Tunic Pharynx with stigmas Endostyle Intestine Gonad Stomach Reproduction and Development Urochordates are monoecious. Gonads are located near the loop of the intestine, and genital ducts open near the atrial siphon. Gametes may be shed through the atrial siphon for external fertilization, or eggs may be retained in the atrium for fertilization and early development. Although self-fertilization occurs in some species, cross-fertilization is the rule. Development results in the formation of a tadpolelike larva with all five chordate characteristics (figure 17.5). Metamorphosis begins after a brief free-swimming larval existence, during which the larva does not feed. The larva settles to a firm substrate and attaches by adhesive papillae located below the mouth. During metamorpho- sis, the outer epidermis shrinks and pulls the notochord and other tail structures internally for reorganization into adult tissues. The internal structures rotate 180°, positioning the oral siphon opposite the adhe- sive papillae and bending the digestive tract into a U-shape. FIGURE 17.5 Tunicate Metamorphosis. Small black arrows show the path of water through the body. In spite of their streamlined shape, cephalochordates are relatively weak swimmers and spend most of their time in a filter-feeding position—partly to mostly buried with their anterior end sticking out of the sand (figure 17.6a). The notochord of cephalochordates extends from the tail to the head, giving them their name. Unlike the notochord of other chordates, most of the cells are muscle cells, making the notochord somewhat contractile. Both of these characteristics are probably adaptations to burrowing. Contraction of the muscle cells increases the rigidity of the notochord by compressing the fluids within, giv- ving additional support when pushing into sandy substrates. Relax- ation of these muscle cells increases flexibility for swimming. Myotomic muscle bundles on either side of the notochord cause undulations that propel the cephalochordate through the water. Longitudinal, ventrolateral folds of the body wall help sta- bilize cephalochordates during swimming, and a median dorsal fin and a caudal fin also aid in swimming. Subphylum Cephalochordata Members of the subphylum Cephalochordata (sef″a-lo-kor-dah′tah) (Gr. kephalo, head + L. chorda, cord) are called lancelets. Lancelets clearly demonstrate the five chordate characteristics, and for that reason they are often studied in introductory zoology courses. The cephalochordates consist of three extant genera, Epig- onichthys, Branchiostoma (amphioxus), and Asymmetron, and about 32 species. They are distributed throughout the world’s oceans in shallow waters that have clean sand substrates. Cephalochordates are small (up to 5 cm long), fishlike ani- mals. They are elongate, laterally flattened, and nearly transparent. miL2218x_ch17_303-312.indd 308 10/21/22 05:06 PM
Final PDF to printer Chordata: Urochordata and Cephalochordata 309 Oral hood upward. Ciliary bands positioned on folded epithelium on the lateral surfaces of gill bars sweep water into the mouth. These ciliary bands beat in waves creating the impression of a rotating wheel, thus the name “wheel organ” (figure 17.6b). Water passes from the pharynx, through pharyngeal slits to the atrium, and out of the body through the atriopore. Food is initially sorted at the cirri. Larger materials catch on cilia of the cirri. As these larger particles accumulate, con- tractions of the cirri throw them off. Smaller, edible particles are pulled into the mouth with water and are collected by cilia on the gill bars and in mucus secreted by the endostyle. As in tunicates, the endostyle is a ciliated groove that extends longitudinally along the midventral aspect of the pharynx. Cilia move food and mucus dor- sally, forming a food cord to the gut. A ring of cilia rotates the food cord, dislodging food. Digestion is both extracellular and intracellular. A diverticulum off the gut, called the midgut cecum, extends anteri- orly. It ends blindly along the right side of the pharynx and secretes digestive enzymes. An anus is on the left side of the ventral fin. Cephalochordates do not possess a true heart. Contractile waves in the walls of major vessels propel blood. Blood contains amoeboid cells and bathes tissues in open spaces. Excretory tubules are modified coelomic cells closely associ- ated with blood vessels. This arrangement suggests active transport of materials between the blood and the excretory tubules. The coelom of cephalochordates is reduced, compared to that of most other chordates. It is restricted to canals near the gill bars, the endostyle, and the gonads. Pharynx Cirri Myomeres Notochord Gill bar Dorsal tubular nerve cord Pharyngeal slits Midgut cecum Dorsal fin Atrium Atriopore Ventral fin Anus Tail (a) Myomeres Oral hood Notochord Cirri Reproduction and Development Cephalochordates are dioecious. Gonads bulge into the atrium from the lateral body wall. Gametes are shed into the atrium and leave the body through the atriopore. External fertilization leads to a bilat- erally symmetrical larva. Larvae are free swimming, but they eventu- ally settle to the substrate before metamorphosing into adults. Wheel organ Pharyngeal slits (b) FIGURE 17.6 Subphylum Cephalochordata. (a) Internal structure of Branchiostoma (amphioxus) shown in its partially buried feeding position. (b) Oral hood and anterior pharyngeal region of Branchiostoma. Science Stock Photography/Science Source 17.3 EVOLUTIONARY CONNECTIONS LEARNING OUTCOMES 1. Describe the relationships between members of the three chordate subphyla. 2. Characterize members of the largest craniate infraphylum, Vertebrata. 3. Hypothesize on the body forms of the basal deuterostome and the common deuterostome ancestor of the three chordate clades. An oral hood projects from the anterior end of cephalochor- dates. Ciliated, fingerlike projections, called cirri, hang from the ven- tral aspect of the oral hood and are used in feeding. The posterior wall of the oral hood bears the mouth opening that leads to a large pharynx. Numerous pairs of pharyngeal slits perforate the pharynx and are supported by cartilaginous gill bars. Large folds of the body wall extend ventrally around the pharynx and fuse at the ventral mid- line of the body, creating the atrium, a chamber that surrounds the pharyngeal region of the body. It may protect the delicate, filtering surfaces of the pharynx from bottom sediments. The opening from the atrium to the outside is called the atriopore (seefigure 17.6a). Evidence supporting the monophyly of the deuterostome clade, and conclusions drawn from that evidence, is presented in chapter 16. Evidence supporting the monophyly of the chordate lineage is derived from a combination of paleontological, developmental, and molecular sources. Conclusions from these data are discussed in the following paragraphs. Ample evidence makes chordate origins from early deutero- stomes certain. All chordates possess pharyngeal slits and display deuterostome developmental characteristics (see figure 8.11). (Unlike Maintenance Functions Cephalochordates are filter feeders. During feeding, they are par- tially or mostly buried in sandy substrates with their mouths pointed miL2218x_ch17_303-312.indd 309 10/21/22 05:06 PM
Final PDF to printer 310 CHAPTER SEVENTEEN other deuterostomes, the coelom of vertebrate chordates develops in a schizocoelous manner. Schizocoelous coelom formation is a derived character that probably evolved during the evolution of large yolky egg cells.) Molecular evidence in the form of 16s and 18s rRNA sequenc- ing, shared HOX genes (see chapter 4), and many phylogenomic studies provide very strong support for deuterostome affinities. (Phylogenom- ics is the study of entire genomes or large portions of genomes rather than the singe-gene sequence studies of phylogenetics.) Molecular and developmental evidence also helps us under- stand the divergence between ambulacrarian and chordate lineages within the deuterostomes. For example, protostomes and deutero- stome share a set of homologous genes that control the develop- ment of the dorsoventral axis of the animal. In protostomes, a growth factor gene (dpp) is active dorsally in the embryo. This gene inhibits the development of the nervous system in the dorsal region of the embryo. Another gene (sog) inhibits dpp ventrally, and a ven- tral nervous system develops. The dpp homolog in deuterostomes is called bone morphogenic protein (bmp) based on its function in vertebrates. Protostome ties to the ambulacraria are evidenced in the activity of bmp. In echinoderms bmp is active dorsally, and in hemichordates bmp has both dorsal and ventral influences. Thus, in ambulacrarians the dorsoventral axis is oriented similarly to that of protostomes. The pharyngeal bars are positioned dorsally in hemi- chordates and fossil echinoderms as compared to their ventral posi- tion in chordates. Recall that enteropneusts possess both dorsal and ventral nerve cords (see figure 16.16), a result of bmp having both dorsal and ventral influences. In chordates, however, bmp is active ventrally, and a dorsal nervous system and ventral pharyngeal bars develop. These developmental changes were undoubtedly important in the divergence of protostome and deuterostome lineages and give clues to the transitional status of ambulacrarian phyla, the ancestral status of Enteropneusta, and the origin of Chordata. Relationships within the Chordata are also being clarified by molecular and paleontological evidence. Molecular evidence, including 18s rRNA sequencing, supports the hypothesis that the cephalochordate lineage is basal to both Urochordata and Craniata (seefigure 17.7). Thus, the basal chordates may have been solitary, weakly mobile, benthic filter feeders that looked and behaved like modern lancelets (seefigure 17.6). Paleontological evidence of early chordate evolution is scanty because soft tissues predominated in Ambulacraria Xenoturbellida Chordata Hemichordata Cephalochordata Enteropneusta* Echinodermata Pterobranchia Urochordata Craniata Buccal apparatus Crown of ciliated feeding tentacles Pharyngeal basket Enlargement of the neural tube forms a three-part brain Sessile and colonial Loss of coelom Tunic Endoskeleton including a cranium Loss of pharyngeal slits Buccal diverticulum Endostyle or thyroid gland Tadpole larva Calcium carbonate endoskeleton Postanal tail Notochord Water vascular system Dorsal tubular nerve cord Difuse epidermal nervous system Tripartite coelom Larvae with ciliary bands Pharyngeal slits Radial cleavage, enterocoelous coelom formation FIGURE 17.7 One Interpretation of Deuterostomate Phylogeny. Developmental and molecular evidence link the echinoderms and hemichordates into the clade Ambulacraria. The Enteropneusta (*) is probably not monophyletic. The dorsal tubular nerve cord, notochord, postanal tail, and endostyle (thyroid gland in adult vertebrates) are important characteristics distinctive of the Chordata. Some of the synapomorphies that distinguish the chordate subphyla are shown. miL2218x_ch17_303-312.indd 310 10/21/22 05:06 PM
Final PDF to printer Chordata: Urochordata and Cephalochordata 311 early chordates. Fossils that have been uncovered have been inter- preted as supporting cephalochordate ancestry. Pikaia (figure 17.8a, 520 million years age [mya]), which was unearthed from Burgess fossil beds (see chapter 8), shows evidence of myomeres and myo- septa, but not pharyngeal slits. Haikouella (figure 17.8b, 530 mya), from fossil beds in China, shows evidence of all chordate character- istics described at the beginning of this chapter. Most authorities now place these species within the Cephalochordata. If Cephalochordata is ancestral to other chordates, the uro- chordate and craniate lineages quickly diverged from these ances- tors. The oldest known urochordate fossil is from South China (Shankouclava, 520 mya) and is similar in body form to modern, solitary ascidians. The loss of the chordate characteristics in adults (i.e., mobile lifestyle, body cavity, notochord, post-anal tail, and tubu- lar nervous system), and the acquisition of a cellulose- containing tunic, must have been founding events in urochordate evolution. Similarly, the early acquisition of a protective cranium surrounding the brain is evidenced in the earliest known craniate fossils (the myl- lokunmingiids, 530–520 mya, see chapter 18). The largest and most successful craniates belong to the infra- phylum Vertebrata. Bony or cartilaginous vertebrae that completely or partially replace the notochord characterize the vertebrates. The development of the anterior end of the nerve cord into a three-part brain (forebrain, midbrain, and hindbrain) and the development of specialized sense organs on the head are evidence of a high degree of cephalization. The skeleton is modified anteriorly into a skull or cra- nium. There are eight classes of vertebrates (see table 17.1). Because of their cartilaginous and bony endoskeletons, vertebrates have left an abundant fossil record. Ancient jawless fishes were common in the Ordovician period, approximately 500 mya (see appendix B). Over a period of approximately 100 million years, fishes became Notochord Myomere (a) Dorsal aorta Notochord Myomere Nerve cord Esophagus Brain Eye Ventral aorta Pharyngeal bar Endostyle Anus Atriopore Intestine (b) FIGURE 17.8 Early Chordates. (a) This drawing is based on a fossil of a 520-million- year-old cephalochordate, Pikaia gracilens, from the Burgess Shale of British Columbia. (b) Haikouella is a cephalochordate fossil (530 million years old) found in fossil beds in China. the dominant vertebrates. Near the end of the Devonian period, approximately 400 mya, terrestrial vertebrates made their appear- ance. Since that time, vertebrates have radiated into most of the earth’s habitats. Chapters 18 through 22 give an account of these events. SUMMARY or solitary urochordates that are also pelagic filter feeders. Some are colonial and bioluminescent. The endostyle of urochordates secretes mucus that is used to trap filtered food within their large pharynx. Urochordates are monoecious with external fertilization and the development of a tadpolelike larva. • The subphylum Cephalochordata includes small, tadpolelike filter feeders that live in shallow marine waters with clean, sandy substrates. Their notochord extends from the tail into the head and is somewhat contractile. Cephalochordates are filter feed- ers. Cilia on the lateral surfaces of gill bars sweep food into the mouth, and food is filtered as water passes though pharyngeal slits and into the atrium. Cephalochordates are dioecious, and fertilization and larval development are external. 17.3 Further Phylogenetic Considerations • Molecular, developmental, and paleontological evidence provide strong support for the conclusion that the Chordata arose from the deuterostome lineage and share common ancestry with the Ambulacraria. Divergence from the ambulacraria probably resulted in the evolution of a cephalochordate-like ancestor about 530 mya. Urochordata and Craniata were derived from that ancestral chordate. The largest and most successful group of cra- niates belong to the infraphylum Vertebrata. 17.1 Evolutionary Perspective • Echinoderms, hemichordates, and chordates share deuterostome characteristics and are believed to have evolved from a common diploblastic or triploblastic ancestor. 17.2 Phylum Chordata • Chordates have five distinctive characteristics. A notochord is a supportive rod that extends most of the length of the animal. Pharyngeal slits are a series of openings between the digestive tract and the outside of the body. The tubular nerve cord lies just above the notochord and expands anteriorly into a brain. A postanal tail extends posteriorly to the anus and is supported by the notochord or the vertebral column. An endostyle functions in filter feeding or a thyroid gland functions as an endocrine organ. • Members of the subphylum Urochordata are the tunicates or sea squirts. Ascidians are sessile filter feeders that attach their saclike bodies to solid substrates. Two siphons permit seawater to circulate through their bodies. Their bodies are covered by a cellulose containing tunic. Appendicularians are pelagic filter feeders that are tadpolelike as adults. Thaliaceans are colonial miL2218x_ch17_303-312.indd 311 10/21/22 05:06 PM
Final PDF to printer 312 CHAPTER SEVENTEEN REVIEW AND APPLY c. Discuss the role of filter feeding in deuterostome evolution. At what point in chordate evolution does feeding become foraging or predatory in nature? d. Compare and contrast metamorphosis in the Echinodermata and the Urochordata. Do any similarities you describe indicate common ancestry of the two groups? 17.3 Evolutionary Connections a. How does dorsoventral patterning in chordate development help inform our understanding of chordate evolution? b. How has our interpretation of the relationship between chor- date subphyla changed because of data from molecular studies? What observations from early studies needed to be reevaluated because of this change in interpretation? 17.1 Evolutionary Perspective a. What important character is shared by deuterostomes (but lost early in the echinoderm lineage)? b. What evidence links echinoderms, hemichordates, and chor- dates to the same evolutionary lineage? 17.2 Phylum Chordata a. How are the five distinctive chordate characteristics represented in the Urochordata and Cephalochordata? b. What evidence of chordate affinities is present in adult tuni- cates? In larval tunicates? If you only could examine an adult, would you be able to identify it as being a chordate? miL2218x_ch17_303-312.indd 312 10/21/22 05:06 PM