Particle Packing
Che5700 陶瓷粉末處理. Particle Packing. Forming: strongly related to particle packing (science and technology) Results from packing: packing density and porosity

Particle Packing
E N D
Presentation Transcript
Che5700 陶瓷粉末處理 Particle Packing • Forming: strongly related to particle packing (science and technology) • Results from packing: packing density and porosity • Factors: particle size and distribution, particle shape, resistance of particles to pressure (deformation; binder effect), flow resistance (friction between particles) For uniform spheres: five different packing arrangements – cubic, orthorhombic, tetragonal, pyramidal, tetrahedral etc. • Different packing density: higher coordination number to higher packing density, theoretical maximum: 74%.
In theory, we can obtain ordered packing of mono-disperse particles; in reality, it is often to get packing as shown above (small range of ordering)
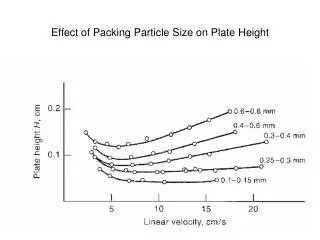
Che5700 陶瓷粉末處理 Packing Density and Pore Size
Che5700 陶瓷粉末處理 Packing Characteristics • Tortuosity o: for cubic packing o= 1.0; tetrahedral packing o= 1.3 • Number of particle contact Nc = 3 (PF) (CN)/( a3) • PF = packing fraction; CN = coordination number • for nonregular packing Nc = 3 (1-)/(a3); since CN ~ / (usually between 6 – 10) • Container wall effect (on packing): insignificant when container dia./particle dia. > 10 • Use two particle sizes, small one can fill into interstice, thus increase packing density
Che5700 陶瓷粉末處理 Furnas Model • In theory, if three kinds particle in packing: • PFmax = PFc + (1- PFc) PFm + (1- PFc)(1- PFm) PFf • f i, w = Wi/W total • Wc = PFcc; medium and fine the same • The small particle size have to be small enough, size ratio > 7, to effectively increase packing density • In industry, often mix two or more particles to get high density packing, to reach densification at lower sintering temperature
圖中直線代表粒子粒徑比值無限大的理論值; • Highest density occurs when small particle fill completely porosity from large particles (volume fraction for fines ~ 26% or porosity from large particles ~26%) • In reality, since the size ratio will not be too large, the highest point of packing density usually moves toward the middle point.
Che5700 陶瓷粉末處理 Packing of Continuous Distribution • E.g. log normal distribution: theoretical calculation shows that, under random packing, larger geometric standard deviation , denser packing (spheres) • Andreasen cumulative distribution (1): usually n = 0.33 – 0.5; experience: 1/n increase, packing density increase • Zheng modified distribution (2): one more parameter, amin
Taken from JS Reed, 1995; often packing density 60-69%; In reality, particles not very spherical, will affect packing density
Results from real particle size distributions, sample: calcined Bayer alumina; it is not very easy to rationalize
Che5700 陶瓷粉末處理 Hindered Packing • Including external and internal factors: • Bridging of particles and agglomerates with rough surface of walls (mechanical vibration [– tap density], lubrication, large force causing particle fracture may improve somewhat;) • Coagulation , adhesion between particles also retard particle motion and hence packing into dense structure • High aspect ratio often produce high porosity • Adsorbed binder molecule also hinder particle movement
Che5700 陶瓷粉末處理 Ordered Structure in Suspension • For monodisperse particle systems: particle interaction + gravity force ordered structure (so called order-disorder phase transition question: a thermodynamic and mechanical equilibrium problem) • Defects : point defect (vacancy), line defect (dislocations), planar defects (grain boundary), volume defects (cracks) • Point defect: can be estimated from thermodynamics; other defects: related to processing • Measurement of ordered domain size: Scherer equation (peak broadening) = FWHM = k/(L cos) = full width at half height; k = constant ~ 0.9
本圖取自TA Ring, 1996; • Measurement of ordered array structure: light diffraction (iridescence) n = 2 d sin can estimate size of structure from diffraction peaks (d)
Sinterbility of Agglomerated Powders • Source: J. Am. Cer. Soc., 67(2), 83-89, 1984 (by FF Lange) • A new concept: Pore coordination number; thermodynamic analysis: pore will disappear only when its coordination number is less than a critical value; • Real system: irregular particle size and shapes & irregular arrangement (packing) • Agglomerates: hard (partially sintered); soft (held by van der Waals forces)
General experiences: soft agglomerates produce better sintering results than hard agglomerates • This author thinks: “particle arrangement” is important • A pore: has its volume, shape and coordination number • R>Rc: pore surface convex; R<Rc: concave surface (those pores are able to disappear)
Theoretical calculation: equal-sized spheres, random packing, pore volume always 0.37 ~ 0.41 (or density: 059 ~ 0.63); for real powder: tap density rarely over 30% of true density • Theoretical calculation: different sized sphere can produce bulk density up to 95%; • Consolidation force to increase bulk density: depend on resistance of particle packing unit to deformation (via particle rearrangement) ; as shear stress increase, agglomerate first to shear apart into their smaller domains, next domain deformation, finally, particle deform or fracture; • Grain growth: a method to reduce pore coordination number; grain growth from mass transport (temperature effect) • If pore growth faster, we may get pores with higher coordination number
Transparent Alumina • Grain size ≦500 nm; residual porosity: negligible (e.g. 0.03%) • Possible methods: (a) Use high sintering temperature (grain growth problem); or (b) through special particle coordination and low temperature sintering (shaping technique or particle size distribution – key: homogeneity; e.g. no agglomerates) • Following data from: J. Am. Cer. Soc. 89(6), 1986-1992, 2006. • Raw material: Al2O3, 99.99% pure, 150-200 nm;
Shaping methods: (a) dry pressing (uniaxial pressing at 200 MPa; cold isostatic pressing CIP at 700MPa (pre-shaped at 30-50 MPa); (b) gel-casting (4-5 wt% monomer); (c) slip casting into porous alumina mold • Binder burnout: 800oC, very small shrinkage (< 0.2%), develop neck, provide strength for Hg intrusion analysis • Mercury porosimetry better than SEM to measure pore size distribution • No large pores (>75 nm): an indication of homogeneity
Pore size distribution: do not change much from green state to intermediate sintering stage; • Homogeneity: poor for uniaxial pressing • Pore size ~ 50 nm ~ 1/3 of particle size
Slip casting provides the best particle coordination: pore size ~ 35 nm ~ 1/5 particle size • Observation: Smaller and larger pore are eliminated at similar rates
(a) slip casting without binder, presintered at 1200oC, then HIP 1170oC, ave. grain size = 0.44 μm • (b) gelcasting, presintered at 1240oC, HIP 1200oC, ave. grain size = 0.53 μm (both densities > 99.9%) • All above data taken from J. Am. Cer. Soc. 89(6), 1985-1992, 2006.