Janots et al., 2007
380 likes | 519 Vues
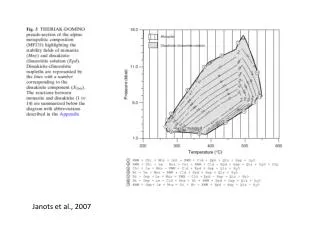
Janots et al., 2007. Spear and Pyle, 2010. Spear, 2010. Spear, 2010. Kelsey et al., 2008.

Janots et al., 2007
E N D
Presentation Transcript
“Coupled dissolution-reprecipitation is a well-established chemical reaction, driven by a minimization in the Gibbs free energy. In this process, a mineral phase, in the presence of a reactive fluid, is replaced either by an altered composition of the same phase or by an entirely new phase (Putnis, 2002)”.
“The most important result of the TEM study is the demonstration of the presence of small (50 nm) Pb-rich domains where large variations in EMP ages occur. We suggest that radiogenic Pb was partially reincorporated in monazite during the recrystallization…” ABSTRACT Electron-microprobe (EMP) U-Th-Pb dating on polyphase and discordant monazites from polymetamorphic granulites of the Andriamena unit (north-central Madagascar) reveals inconsistent chemical ages. To explain these drastic variations, transmission electron microscopy (TEM) foils were prepared directly from thin sections by using the focused ion beam technique. The most important result of the TEM study is the demonstration of the presence of small (;50 nm) Pb-rich domains where large variations in EMP ages occur. We suggest that radiogenic Pb was partially reincorporated in monazite during the recrystallization at 790 Ma. Because the excited volume of EMP is ;4 mm3, U-Th-Pb dating yielded various apparent older ages without geological significance. In addition, TEM analysis of the foils revealed the presence of an ;150-nm-wide amorphous zone along the grain boundary of monazite and its host quartz. This Fe-Si-Al–rich phase may have formed as a result of fluid activity at 500 Ma, and the phase’s amorphous state may be due to the irradiation from U and Th decay in the monazite. This demonstrates for the first time the enormous potential of the TEM investigations on site-specific specimens prepared with the focused ion beam technique for the interpretation of geochronological data.
B. Budzyn (2009) M-21
Assemblage: monazite, muscovite, albite, amorphous SiO2 Reagents: CaF2 Na2Si2O5 • Experimental conditions • 4.5 kbar, 450°C for a • Duration: 16 days. Experiment: hydrothermal apparatus See: Budzyn (2009)
Starting monazite The monazite chosen for the experiment was taken from a heavy-mineral sand deposit at Cumuruxatiba, Bahia State, Brazil D. Moderately rounded, semi-euhedral, relatively transparent, inclusion-free, 100 – 500 mm, amber-colored grains. The monazite grains were hand-picked out of the heavy mineral sand, crushed to 50 – 150 mm size fragments and then washed in ethanol in an ultrasonic bath. ThO2: 7-8 wt %, UO2: 0.5-0.75 wt%
Assemblage: monazite, muscovite, albite, amorphous SiO2 Reagents: CaF2 Na2Si2O5 • Experimental conditions • 4.5 kbar, 450°C for a • Duration: 16 days. See: Budzyn (2009)
M-21 5m
M-X ThO2= ~7wt% ThO2= ~8wt% 5m
M-Z 5m
M-21 Bkg-1 Bkg-3 Bkg-2 5m
Compiled and modified by G. Dumond from Hoffman (1988), Tella et al. (2000), Ross (2002), Hajnal et al. (2005), van Breeman et al. (2005; 2007), Rainbird and Davis (2007), Berman et al. (2007), & http://www.lithoprobe.ca/transects