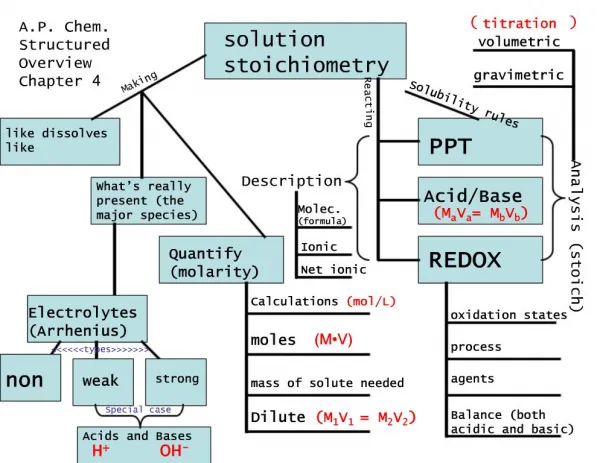
Solution Stoichiometry
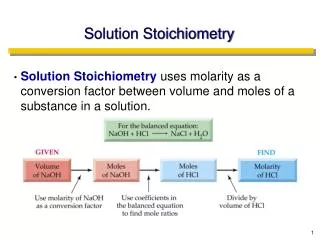
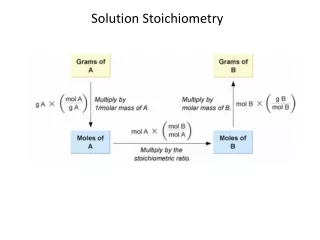
Solution Stoichiometry. Solution Stoichiometry calculations involve chemical reactions that take place in solution. Balanced Chemical reactions are written in terms of moles of reactants and products.

Solution Stoichiometry
E N D
Presentation Transcript
Solution Stoichiometry • Solution Stoichiometry calculations involve chemical reactions that take place in solution. • Balanced Chemical reactions are written in terms of moles of reactants and products.
Molarity relates moles of solute to volume of solutions. Thus, a measured volume of a solution provides a simple method of measuring the moles of reactants. • *We use molarity and volume to convert to moles instead of mass and molar mass. • Example: How many moles are in 15mls of 0.76M KCl? • 15mls 1L 0.76 moles 1000mls 1 liter = 0.011 moles KCl
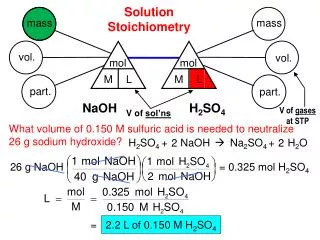
Solution stoichiometry will give you volumes of solutions of reactants and ask how many grams of a precipitate will form. • Example: Silver nitrate is added to 25.0 mls of a 0.50M KCl solution until no more precipitate forms. What mass of AgCl will be formed? • Step 1 – Write the balanced chemical reaction. • AgNO3(aq) + KCl(aq) AgCl(s) + KNO3(aq) • Step 2 – Set up dimensional analysis solution. 0.50mols KCl 1 mol AgCl 143.35g 25.0mls 1 L 1000ml 1 L 1mol KCl 1 mol AgCl = 1.79 grams AgCl should form.
It can also allow you to determine what volume of a reactant is needed to react away a given volume of another reactant. • Example - What volume of 0.50M KCl is required to react away all the silver ions in 45mls of 0.35M AgNO3? • AgNO3(aq) + KCl(aq) AgCl(s) + KNO3(aq) 0.35mol AgNO3 1mol KCl 45mls AgNO3 1 L 1 L KCl 1mol AgNO3 1000mls 0.5mols KCl 1 L 0.0315 Liters = 31.5 mls of 0.50M KCl is required