Optimizing QTc Prolongation Detection in Oncology Trials: A Novel Approach
This study presents an optimized design for QTc prolongation detection during oncology drug trials. QTc prolongation is a critical biomarker for cardiac risk, particularly in evaluating new anti-cancer medications. Utilizing a population pharmacokinetic model, we analyze circadian rhythm effects on QTc intervals, assess empirical ECG measurement times, and calculate detection power for QTc changes. Findings aim to enhance future trial designs, ensuring robust QTc evaluations and improving patient safety.


Optimizing QTc Prolongation Detection in Oncology Trials: A Novel Approach
E N D
Presentation Transcript
Optimal design and QT-prolongation detection in oncology studies Sylvain Fouliard & Marylore Chenel Department of clinical PK, Institut de Recherches Internationales Servier, Courbevoie, FrancePODE Meeting – Berlin - 11th June 2010
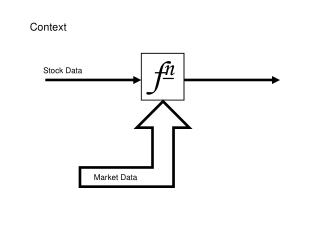
CONTEXT (1) • QT prolongation, a biomarker of Torsade de Pointes. • QT measured on ECG, then corrected. • Circadian rythm in QT/QTc data • Usually mandatory QT/QTC study performed in healthy volunteers at supratherapeutic dose • Guidelines: mean QTc effect > 5ms
CONTEXT (2) • New anti-cancer drug in clinical development - QTc-prolongation = class effect ? • Development of anticancer drugs: patients only • 2 phase I studies: • PK data available population PK model • No QT data available • 2 ongoing phase I/II studies - QTc-prolongation assessment: ECG measurement times already decided without optimization (=empirical design) • Internal QT database in HV (wo drug) population circadian QTc model available
CONTEXT (3)EMPIRICAL DESIGN Inclusion D1 D2 D4 D14 D22 Treatment No Treatment ECG Dose • 2 phase I clinical trials: n = 60 + 40 (=100) patients • Dose regimen : 14 days on / 7 days off, BID administration (4h apart) • 14 ECG measurements per patient • Same measurement times for all patients ECG times : Inclusion D1 D2 D4 D14 D22 0 0, 1.5, 0, 1.5h 0, 1.5h 0, 1.5h 0, 1.5h 4, 5.5, 8 h
OBJECTIVES • Evaluate the Empirical design for ECG Times. • Calculate the Power of detection of a QTc effect in the on going phase I/II studies. • Optimize the ECG Measurement Times for future studies.
MATERIALS & METHODS (1) POPULATION QTc MODEL WITHOUT DRUG Assumption: same model to describe the circadian rhythm in QTc in HV and in patients • Model building dataset: 2 thorough QT/QTc studies • 62 + 87 (=149) healthy volunteers • QT data without drug • Fredericia correction: QTc = QT * HR-0.33 • Model characteristics • - poly-cosine model [1] • - IIV on all parameters • - Additive error model • Software (estimation method): • - NONMEM VI (FOCEI) • Criteria : LRT • Evaluation: GOF, RSE, VPC QTc (ms) Median 5% - 95 % CI Observations … Time (h) [1] Piotrovsky, V. “Pharmacokinetic-pharmacodynamic modeling in the data analysis and interpretation of drug-induced QT/QTc prolongation” (2005)
MATERIALS & METHODS (2) POPULATION QTc MODEL WITH DRUG EFFECT • Assumptions: • Same model to describe the circadian rhythm in QTc in HV and in patients • Concentration proportional drug effect on Mesor • QTc-prolongation is measured by : • Max QTc-prolongation at Cmax (PKPD model)
MATERIALS & METHODS (3) POPULATION PK MODEL F Ka Q3 Q2 Periph. 1 (V2) Central (V1) Periph. 2 (V3) CL • Model building dataset: 2 phase 1 studies • - 14 patients, IV multiple doses, oral single dose • - 35 patients, oral multiple doses • Model characteristics: • - 3-compartments model • - First order absorption and elimination • - IIV on Ka, F, CL, V1, V2 • - Combined error model • Software (estimation method): NONMEM VI (FOCEI) • Criteria : LRT • Evaluation: GOF, RSE, VPC more
MATERIALS & METHODS (5)CALCULATION OF FISHER INFORMATION MATRIX PK model PK parameters (not estimated) QTc model without treatment Mesor, 3 Cosine amplitude terms 3 Cosine Lagtime (estimated) Sequential pop PKPD modelling Drug effect γ (estimated) QTc model under treatment 8 parameters + Additive error QTM0, QTA1, QTA2, QTA3, QTL1, QTL2, QTL3, γ
MATERIALS & METHODS (6)EVALUATION OF THE EMPIRICAL DESIGN To find the range of relevant γ values corresponding to a range of relevant QTc prolongations Range of relevant QTc-prolongation values [1 ms, 100ms] Range of relevant γ values [0.01, 1] • Calculation of the population Fisher Information Matrix • Parameters of QTc model without drug • γ = {0. 01, 0.02, 0.05, 0.1, 0.15, 0.2, 0.3, 0.4, 0.5, 0.8, 1} • IIV on γ = 30 % • Output results: • SE, RSE, DET (determinant of the population FIM)
MATERIALS & METHODS (7)POWER DETECTION OF DRUG EFFECT For each value of γ, SE(γ) is computed from FIM Wald test is performed, with a 5 % type I error. - Null hypothesis H0 : no QTc effect of the drug, 0 = 0 - Alternative hypothesis H1: QTc effect of the drug, 0 > 0 Then power is computed from the type II error β. Power = 1- β.
MATERIALS & METHODS (8)ECG DESIGN OPTIMIZATION • Design characteristics : • - 1 group of patients • - = 0.05, 30 % IIV • - Same days* & number of measurement per day* as the empirical design, design domain = [0-10h] for D1 • = [0-8h] for each other ECG measurement day • Output results : • Optimal ECG times • SE, RSE, DET (determinant of the population FIM) • * 5 ECG on D1, 2 ECG on D2, 2 ECG on D4, 2 ECG on D14, 2 ECG on D22
MATERIALS & METHODS (9) • Software: • - PopDes [2], version 3.0 under MATLAB • Design options: • Local, Population, Univariate (design variable = ECG measurement time only, i.e. PK fixed) • Optimisation method: Fedorov Exchange • Criteria : D-Optimality [2] Gueorguieva, K. Ogungbenro, G. Graham, S. Glatt, and L. Aarons. A program for individual and population optimal design for univariate and multivariate response pharmacokinetic and pharmacodynamic models. Comput. Methods Programs Biomed. 86(1): 51-61 (2007)
RESULTS (1)EMPIRICAL DESIGN EVALUATION (1) • Whatever the values (i.e. drug effect), there is low impact on the RSEs of baseline QTc model parameters. • SE() increases with ; RSE is below 20 % for > 0.05 (QTc-prolongation of 5 ms).
RESULTS (2)EMPIRICALDESIGN EVALUATION (2) RSE of QTc model parameters for a drug effect () of 0.05 (corresponding to a QTc prolongation of about 5 ms). • The RSEs of QTc model parameters are always lower than 20% for fixed effects, except for QTA1, for which there are around 25%.
RESULTS (3)POWER DETECTION OF DRUG EFFECT Power of drug effect detection versus value (drug effect size) • Power > 90 % for > 0.02, corresponding to a 2 ms average QTc-prolongation.
RESULTS (4)ECG TIME OPTIMIZATION (1) RSE comparison for each parameter of the empirical and the optimal designs Empirical design (Det = 2.37 x 1040) Optimal design (Det = 2.22 x 1064) Sampling times : D1 D2 D4 D14 D22 Phase I/IIdesign 0, 1.5, 4, 5.5, 8h 0, 1.5h 0, 1.5h 0, 1.5h 0, 1.5h Optimized design 4, 8, 8.2, 8.8, 9.6h 1.5, 5.6h 3.8, 5.2h 0, 0.6h 1, 1.5h • The optimal design is better than the empirical one, especially for QTA1.
CONCLUSIONS INTERESTS & LIMITS • This work reassured us on the capability of the empirical design to detect any potential drug effect. • The empirical design should allow an accurate estimation of the parameters of the QTc model under treatment. • Several assumptions have been made clinicians not ready yet to have an adaptive design within a study.
CONCLUSIONS (2) NEXT STEPS • Assumptions made will be challenged with first clinical data coming. • PK model • QTc baseline model parameter values • Linear drug effect • Optimization of the ECG measurement times with different clinical constraints (days, times, number of group, doses, number of measurements) for further studies. • Interest in having an integrated tool for estimation and optimization.
Sylvain Fouliard pharmacometrician at Servier ACKNOWLEDGMENT France Mentré
RESULTSMODEL BUILDING Population PK model Parameter estimates and RSE of the population PK model Parameter Back
RESULTSMODEL EVALUATION Population PK model Visual predictive checks Normalized dose Normal scale Log scale Time (h) Median 5% - 95 % CI Observations … Back
Observed Values compared to Simulated Confidence IntervalObserved Values compared to Simulated Confidence Interval RESULTSMODEL EVALUATION CI Obs below CI (%) Obs in CI (%) Obs above CI (%) MEDIAN 61.1 . 38.8 [P1-P99] 1.7 97.6 0 [P5-P95] 6.1 91.7 2.2 [P10-P90] 10.4 85.3 4.3 [P25-P75] 26.4 60.2 13.4 Population PK model Numerical predictive checks Back
RESULTSMODEL BUILDING Baseline poly-cosine QTc model Parameter estimates and RSE of the baseline poly-cosine QTc model Parameter Back
RESULTSMODEL EVALUATION QTc (ms) Median 5% - 95 % CI Observations … Time (h) Baseline poly-cosine QTc model Visual predictive checks Back
Observed Values compared to Simulated Confidence IntervalObserved Values compared to Simulated Confidence Interval RESULTSMODEL EVALUATION CI Obs below CI (%) Obs in CI (%) Obs above CI (%) MEDIAN 49.2 . 50.8 [P1-P99] 0.6 97.6 1.8 [P5-P95] 3.8 90.6 5.6 [P10-P90] 9.4 80.6 10 [P25-P75] 23.3 51.9 24.8 Baseline poly-cosine QTc model Numerical predictive checks Back
CONTEXT (1’) • P wave: auricular depolarisation • QRS complex: ventricular depolarisation • T wave: auricular repolarisation
CONTEXT (1’’) QTc vs. RR • Relationship between QT and RR (=60/HR1000) • Compare QT before and after treatment, once QT is corrected for HR (QTc) QT vs. RR