The Solution Process
The Solution Process. Chemistry Mrs. Coyle. Solution. A homogeneous mixture. One phase. Stainless Steel (Fe, Cr, Ni). Solute, Solvent. Solute —the substance being dissolved. Example: When you dissolve CuCl 2 in water, CuCl 2 is the solute.

The Solution Process
E N D
Presentation Transcript
The Solution Process Chemistry Mrs. Coyle
Solution • A homogeneous mixture. • One phase. Stainless Steel (Fe, Cr, Ni)
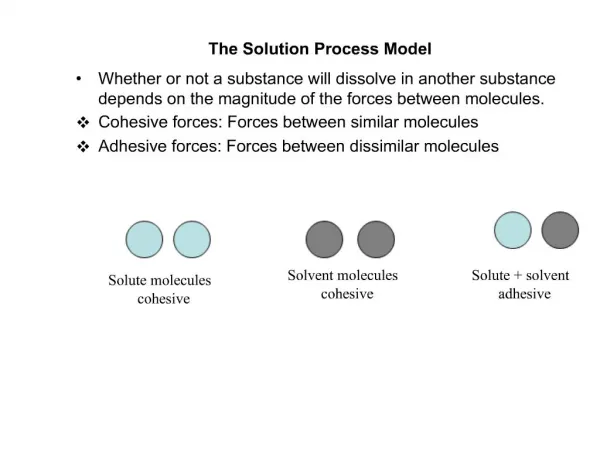
Solute, Solvent • Solute—the substance being dissolved. • Example: When you dissolve CuCl2 in water, CuCl2 is the solute. • Solvent- the substance that dissolves the solute. • Example: water
Aqueous Solution • A solution that has water as the solvent. • Possible substances that can dissolve in water: • Ionic compounds • Polar covalent compounds
Solvation • The surrounding of solute particles by solvent particles.
Dissociation of Ionic Compounds • the process by which an ionic compound separates into its ions as it dissolves.
Movie Clip- Dissociation of Salt in Water http://www.youtube.com/watch?v=EBfGcTAJF4o
Polar ethanol molecule is dissolved by the polar water molecule. Ethanol remains intact.
Will ionic compounds conduct electric current when dissolved in water? • Yes • Why?
Electrolytes and Non-electrolytes • Electrolytes: conduct an electric current when in the molten state or in aqueous solution. Ionic compounds • Non-electrolytes do not conduct a current. Usually molecular compounds
Do all electrolytes conduct electricity to the same degree? • Weak electrolytes: partially ionize in water and conduct electricity in solution poorly (ex. Ammonia) • Strong electrolytes: fully ionize in water and conduct electricity in solution strongly(ex. NaCl).
Hydrate: • A crystalline compound in which the ions are attached to one or more water molecules.
Example: • CuSO4•5H2O • copper(II) sulfate pentahydrate
mono- 1 di- 2 tri- 3 tetra- 4 penta- 5 hexa- 6 hepta- 7 octa- 8 nona- 9 deca- 10 Prefixes for naming Hydrates
Analyzing Hydrates • Simulation click on the link below: • http://www.chem.iastate.edu/group/Greenbowe/sections/projectfolder/flashfiles/stoichiometry/empirical.html
Problem • Calculate the percent by mass of water in washing soda (Na2CO3.10 H2O) • % mass of H2O = MM water x100 % MM Hydrate • Answer: 62.9%
Efflorescent Hydrates • Hydrates that have high vapor pressures compared to water. • When the vapor pressure of the surrounding is lower than the vapor pressure of the hydrate, the hydrate will lose its water; it effloresces.
Hygroscopic Hydrates and Dessicants • Hydrates that have a low vapor pressure compared to water. • These hydrates can absorb water from the air. • These can be used as dessicants (ex. CaSO4).
Deliquescent • Materials that absorb so much water that they will become wet (form solutions). Ex. NaOH.
Colloids and Suspensions • Heterogeneous Mixtures
Suspension • A mixture whose particles are temporarily suspended in a medium, but eventually settle down. • Particle size>100nm • Ex: dust in air.
Colloid • A mixture whose particles (of size ~1 to ~100nm) are dispersed through a continuous medium. (The word colloid means “glue-like”) • Heterogeneous because there are distinct phases. • Tyndall Effect: Scattering of light.
Types of Colloids • Aerosol: liquid or solid in dispersed in gases (fog, smoke). • Foam: gas in liquid(whipped cream). • Emulsion: both substances are liquids (mayonnaise). • Sol: solid in liquid (jelly)