Aqueous Solution Chemistry Process
Learn about solvation, dissociation, electrolytes, hydrates, and heterogeneous mixtures. Discover how different substances dissolve in water and conduct electricity. Explore the simulation for analyzing hydrates.

Aqueous Solution Chemistry Process
E N D
Presentation Transcript
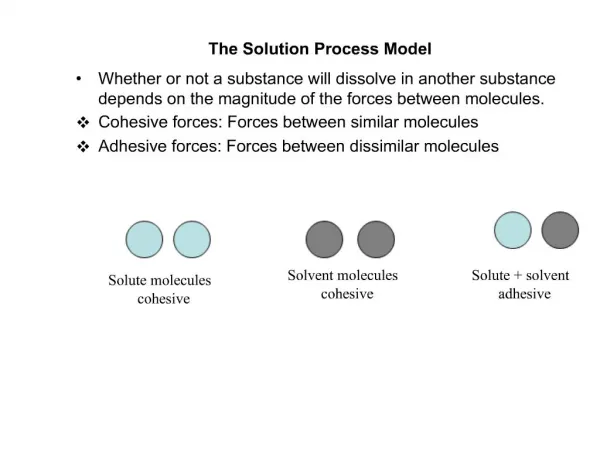
The Solution Process Chemistry Mrs. Coyle
Solution • A homogeneous mixture. • One phase. Stainless Steel (Fe, Cr, Ni)
Solute, Solvent • Solute—the substance being dissolved. • Example: When you dissolve CuCl2 in water, CuCl2 is the solute. • Solvent- the substance that dissolves the solute. • Example: water
Aqueous Solution • A solution that has water as the solvent. • Possible substances that can dissolve in water: • Ionic compounds • Polar covalent compounds
Solvation • The surrounding of solute particles by solvent particles.
Dissociation of Ionic Compounds • the process by which an ionic compound separates into its ions as it dissolves.
Movie Clip- Dissociation of Salt in Water http://www.youtube.com/watch?v=EBfGcTAJF4o
Polar ethanol molecule is dissolved by the polar water molecule. Ethanol remains intact.
Will ionic compounds conduct electric current when dissolved in water? • Yes • Why?
Electrolytes and Non-electrolytes • Electrolytes: conduct an electric current when in the molten state or in aqueous solution. Ionic compounds • Non-electrolytes do not conduct a current. Usually molecular compounds
Do all electrolytes conduct electricity to the same degree? • Weak electrolytes: partially ionize in water and conduct electricity in solution poorly (ex. Ammonia) • Strong electrolytes: fully ionize in water and conduct electricity in solution strongly(ex. NaCl).
Hydrate: • A crystalline compound in which the ions are attached to one or more water molecules.
Example: • CuSO4•5H2O • copper(II) sulfate pentahydrate
mono- 1 di- 2 tri- 3 tetra- 4 penta- 5 hexa- 6 hepta- 7 octa- 8 nona- 9 deca- 10 Prefixes for naming Hydrates
Analyzing Hydrates • Simulation click on the link below: • http://www.chem.iastate.edu/group/Greenbowe/sections/projectfolder/flashfiles/stoichiometry/empirical.html
Problem • Calculate the percent by mass of water in washing soda (Na2CO3.10 H2O) • % mass of H2O = MM water x100 % MM Hydrate • Answer: 62.9%
Efflorescent Hydrates • Hydrates that have high vapor pressures compared to water. • When the vapor pressure of the surrounding is lower than the vapor pressure of the hydrate, the hydrate will lose its water; it effloresces.
Hygroscopic Hydrates and Dessicants • Hydrates that have a low vapor pressure compared to water. • These hydrates can absorb water from the air. • These can be used as dessicants (ex. CaSO4).
Deliquescent • Materials that absorb so much water that they will become wet (form solutions). Ex. NaOH.
Colloids and Suspensions • Heterogeneous Mixtures
Suspension • A mixture whose particles are temporarily suspended in a medium, but eventually settle down. • Particle size>100nm • Ex: dust in air.
Colloid • A mixture whose particles (of size ~1 to ~100nm) are dispersed through a continuous medium. (The word colloid means “glue-like”) • Heterogeneous because there are distinct phases. • Tyndall Effect: Scattering of light.
Types of Colloids • Aerosol: liquid or solid in dispersed in gases (fog, smoke). • Foam: gas in liquid(whipped cream). • Emulsion: both substances are liquids (mayonnaise). • Sol: solid in liquid (jelly)