Heat Engines and Efficiency in Thermodynamics
Explore the principles of heat engines, steam turbines, and efficiency in thermodynamics, including real processes and the second law. Learn about the conversion of thermal energy into mechanical work and the application of conservation of energy.

Heat Engines and Efficiency in Thermodynamics
E N D
Presentation Transcript
Second Law I • Real processes often make sense in only one direction in time. • The second law of thermodynamics states this: Heat flows naturally from a hot object to a cold object; heat will not flow spontaneously from a cold object to a hot object.
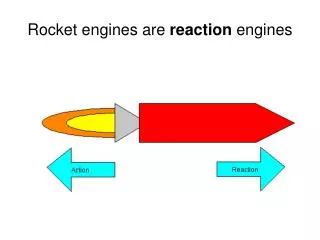
Heat Engine • A device that changes thermal energy into mechanical work is a heat engine. • Requires two temperature reservoirs • Takes heat in and exhausts heat out • Conservation of energy applies. • |QH| = |QL| + |W|
Steam Engine • Steam is produced setting the high temperature. • Expanding steam pushes a piston doing work. • Steam is condensed setting the low temperature.
Steam Turbine • A steam turbine replaces a piston with a paddlewheel. • Most power plants use steam turbines. • Fuel is used to make steam • Coal • Oil • Natural gas • Uranium
Efficiency • The efficiency of a heat engine compares the work done to the heat into the engine. • Measures fuel and work • Efficiency also can compare heat in and out.
An auto engine has an efficiency of 20% and produces 23 kW of mechanical power. How much heat is discharged per second? The efficiency is 0.20. The output work in 1 s is W = 23 kJ. Find the input heat. QH = W / e = 115 kJ Find the output heat. QL / QH = 1 – e QL = QH (1 – e) = 92 kJ DQL / Dt = 92 kW Exhaust
Second Law II • Efficiency can only equal 1 when QL = 0. • Perfect conversion to mechanical work • Not possible with real systems • The second law of thermodynamics can also read: No device is possible whose sole effect is to transform a given amount of heat into work.