Polymers
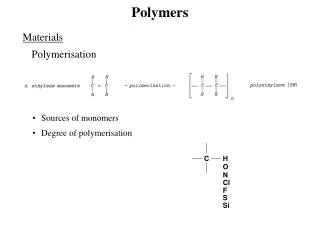
Polymers. What Are Polymers?. Large molecule that consists of lots of small repeating subunits Subunits are called monomers Polymers are formed when monomers are linked together is a process called polymerization. Addition Polymers. Polymers are linked through addition reactions

Polymers
E N D
Presentation Transcript
What Are Polymers? • Large molecule that consists of lots of small repeating subunits • Subunits are called monomers • Polymers are formed when monomers are linked together is a process called polymerization
Addition Polymers • Polymers are linked through addition reactions • Three stages in synthesis • Initiation • Propagation • Termination
Polyethylene • Polymer made from ethene monomers
Polyethylene • Polymer made from ethene monomers • Short form notation:
Polypropene • Made from propene monomers
Polypropene • Made from propene monomers • Used in rope and carpets • Short form notation:
Polyvinyl Chloride (PVC) • Made with chloroethene (vinyl chloride) monomers
Polyvinyl Chloride (PVC) • Made with chloroethene (vinyl chloride) monomers • Used in electrical wire insulation, plastic piping, coating on fabrics • Short form notation:
Polystyrene • Made from (styrene) phenylethene • Very commonly used
Substituted Groups • Change the properties of polymer • Teflon monomer: tetrafluroethane • Plexiglass monomer: methyl propenoate
Crosslinking • Occurs when links can be formed in 2 directions • Diene monomers can form bridges between chains • Diene doesn’t have to be main monomer • Increases polymers’ strength
Plastic Properties • Substituted ethene (vinyl) monomers considered “plastics” • Saturated alkanes • Very stable • Unreactive • Flexible and mouldable solids or liquids • Weak van der Waals forces • Softened by heating • Disrupts forces
Condensation Polymers • Monomer units are joined by condensation reactions • Monomer units must have functional groups at each end
Polyesters • Formed between dicarboxylic acid and diol
Polyesters • Formed between dicarboxylic acid and diol
Polyamides • Formed between dicarboxylic acid and diamine
Polyamides • Formed between dicarboxylic acid and diamine